i) K+ has three energy levels while Na+ has only two.
ii) Mg2+ nucleus has 12 protons attracting 10e- while Na+ has 11 protons attracting 10e- hence Mg2+ radius shrinks more.
johnmulu answered the question on April 10, 2017 at 08:11
- The information in the table below relates to elements in the same group of the periodic table. Study it answer the question that follows:
(Solved)
The information in the table below relates to elements in the same group of the periodic table. Study it answer the question that follows:
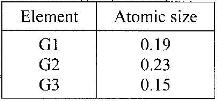
Which element has the highest ionization energy? Give a reason.
Date posted: April 10, 2017. Answers (1)
- The grid given below represents part of the periodic table. Study it and answer the questions that follow. (The letters do not represent the actual symbol of the elements)(Solved)
The grid given below represents part of the periodic table. Study it and answer the questions that follow. (The letters do not represent the actual symbol of the elements)
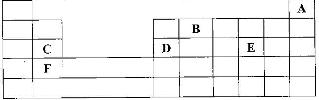
i) What name is given to the group of elements to which C and F belong?
ii) Which letter represents the element that is the least reactive?
iii) What type of bond is formed when B and E react? Explain
iv) Write the formula of the compound formed when element D and oxygen gas react
Date posted: April 10, 2017. Answers (1)
- The table below gives the energy required to remove the outermost electron for some group I elements(Solved)
The table below gives the energy required to remove the outermost electron for some group I elements
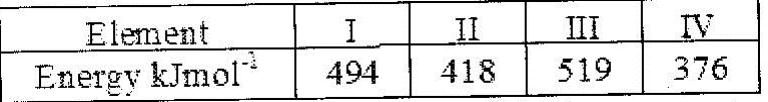
Arrange the elements in order of their reactivity starting with the most reactive.
Date posted: April 7, 2017. Answers (1)
- The grid below shows part of a periodic table. The letters do not represent the actual symbols of the elements.(Solved)
The grid below shows part of a periodic table. The letters do not represent the actual symbols of the elements.
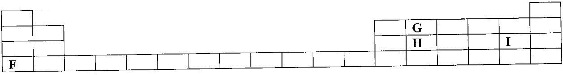
![]() a) Select the;
a) Select the;
i) Element which has the largest atomic radius.
ii) Most reactive non-metal.
Date posted: April 7, 2017. Answers (1)
- Study the table below and answer the questions that follow:(Solved)
Study the table below and answer the questions that follow:
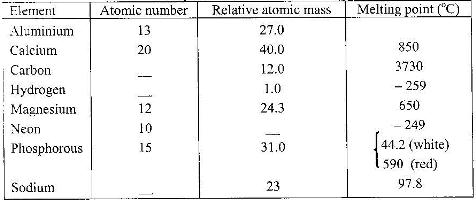
a) Complete the table by filling in the missing atomic numbers and atomic mass
b) Write the electron arrangement for the following ions: (i) Ca+ (ii) P3-
c) What is the melting point of hydrogen in Kelvin?
d) Which of the allotropes of phosphorous has a higher density? Explain.
e) The mass numbers of three isotopes of magnesium are 24, 25 and 26.
What is the mass number of the most abundant isotope of magnesium? Explain.
f) Give the formula of the compound formed between aluminium and carbon.
g) Explain the difference in the melting points of magnesium and sodium.
Date posted: April 7, 2017. Answers (1)
- The table below gives the atomic numbers of elements W, X, Y and Z. The letters do not represent the actual symbols of the elements.(Solved)
The table below gives the atomic numbers of elements W, X, Y and Z. The letters do not represent the actual symbols of the elements.
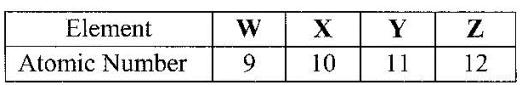
a) Which one of the elements is least reactive? Explain.
b) (i) Which two elements would react most vigorously with each other?
(ii) Give the formula of the compound formed when the elements in b (i) react.
Date posted: April 7, 2017. Answers (1)
- The table below gives information on four elements represented by letters K, L, M and N. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.(Solved)
The table below gives information on four elements represented by letters K, L, M and N. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.
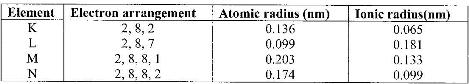
a) Which two elements have two similar properties? Explain.
b) What is the most likely formula of oxide of L?
c) Which element is a non-metal? Explain
d) Which one of the elements is the strongest reducing agent? Explain.
e) Explain why ionic radius of N is less than that of M.
f) Explain why the ionic radius of L is bigger than its atomic radius.
Date posted: April 7, 2017. Answers (1)
- The table below is part of the periodic table. The letters are not the actual symbols of the elements. Study it and answer the questions that follow.(Solved)
The table below is part of the periodic table. The letters are not the actual symbols of the elements. Study it and answer the questions that follow.
a) Select an element which is stored in paraffin in the laboratory
b) How do the ionic radii of E and I compare? Explain
Date posted: April 7, 2017. Answers (1)
- The table below shows behavior of metals R, X, Y and Z. Study it and answer the questions that(Solved)
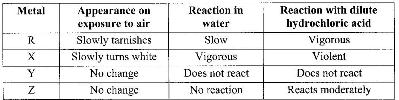
The table below shows behavior of metals R, X, Y and Z. Study it and answer the questions that follow:

a) Arrange the metals in the order of reactivity starting with the most reactive.
b) Name a metal which is likely to be: (i) X (ii) Y
Date posted: April 7, 2017. Answers (1)
- a) What is meant by the term radial?
b) The table below contains atoms that form common radicals. Complete the table to show radicals formed from various atoms
(Solved)
a) What is meant by the term radial?
b) The table below contains atoms that form common radicals. Complete the table to show radicals formed from various atoms
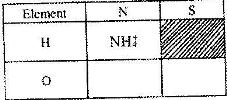
Date posted: April 7, 2017. Answers (1)
- Use the part of the periodic table given below to answer the questions that follow. (letters are not the actual symbols of elements.)(Solved)
Use the part of the periodic table given below to answer the questions that follow. (letters are not the actual symbols of elements.)
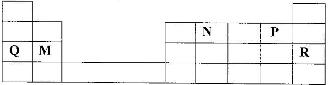
a) Identify the element that forms giant covalent structures.
b) Identify one element that does not form compounds
c) Write the formula for the nitride of M.
Date posted: April 7, 2017. Answers (1)
- The table below gives the number of electrons, protons and neutrons in substances X, Y and Z. Study it and answer the questions that follow.(Solved)
The table below gives the number of electrons, protons and neutrons in substances X, Y and Z. Study it and answer the questions that follow.

a) Which letter represents an ion?
b) Which of the substances are isotopes? Give reason.
Date posted: April 7, 2017. Answers (1)
- The table below shows the number of valence electrons of the element P, Q and R.(Solved)
The table below shows the number of valence electrons of the element P, Q and R.

a) Explain why P and R would not be expected to form a compound.
b) Write an equation to show the effect of heat on the carbonate of R.
c) Write the formula for the most stable ion of Q
Date posted: April 7, 2017. Answers (1)
- The table below shows the relative atomic masses and the percentage abudance of the isotopes L1 and L2 of element L.(Solved)
The table below shows the relative atomic masses and the percentage abudance of the isotopes L1 and L2 of element L.

Calculate the relative atomic mass of element L.
Date posted: April 7, 2017. Answers (1)
- Use the information in the table below to answer the questions that follow.
(The letters do not represent the actual symbols of the elements)
(Solved)
Use the information in the table below to answer the questions that follow.
(The letters do not represent the actual symbols of the elements)

a) Which two letters represents the same element? Give reason.
b) Give the number of neutrons in an atom of element D.
Date posted: April 7, 2017. Answers (1)
- Complete the table below.(Solved)
Complete the table below.

Date posted: April 7, 2017. Answers (1)
- The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.(Solved)
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.

a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
Date posted: April 7, 2017. Answers (1)
- The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.(Solved)
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.

a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
Date posted: April 7, 2017. Answers (1)
- The set-up below was used to investigate the products of burning biogas (methane). Study it and answer the questions that follow.(Solved)
The set-up below was used to investigate the products of burning biogas (methane). Study it and answer the questions that follow.

a) What product will be formed in test-tube Y?
b) State and explain the observations which will be made in Z.
Date posted: April 7, 2017. Answers (1)
- A student used the set up shown in the diagram below in order to study the reactions of some metals with steam. The experiment was carried out for ten minutes.(Solved)
A student used the set up shown in the diagram below in order to study the reactions of some metals with steam. The experiment was carried out for ten minutes.

a) What observation would be made if gas F is ignited?
b) When the experiment was repeated using iron powder instead of magnesium ribbon, very little gas F was obtained.
i) Give a reason for this observation.
ii) What change in the conditions of the experiment should the student have made in order to increase the volume of gas F produced?
Date posted: April 7, 2017. Answers (1)