-
Study the scheme below hence answer the questions that follow.
(Solved)
Study the scheme below hence answer the questions that follow.
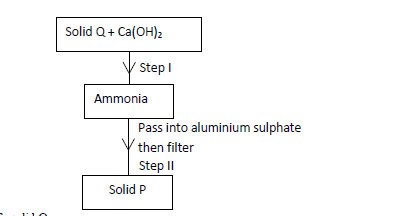
(a) Identify solid Q.
(b) Write an ionic equation for the reaction in Step II.
(c) State the condition necessary for Step I.
Date posted:
March 28, 2019
.
Answers (1)
-
Name the method of separation that can most suitably be used to separate the following mixtures.
(a) Gasoline from petroleum
(b) Benzoic acid and potassium carbonate
(c) Oil...
(Solved)
Name the method of separation that can most suitably be used to separate the following mixtures.
(a) Gasoline from petroleum
(b) Benzoic acid and potassium carbonate
(c) Oil from cashew nuts
Date posted:
March 28, 2019
.
Answers (1)
-
Study the set-up below and answer the questions that follow.
After sometimes, the cotton wools X, Y and Z changed color in turn.
(a) What were the...
(Solved)
Study the set-up below and answer the questions that follow.
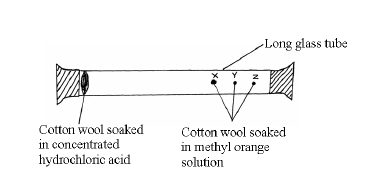
After sometimes, the cotton wools X, Y and Z changed color in turn.
(a) What were the colour changes?
(b) Which cotton wool changed colour first?
(c) Explain why the cotton wools did not change colour at the same time.
Date posted:
March 28, 2019
.
Answers (1)
-
A current of 2.5A was passed through a cell containing N2+ ions for 25 minutes. The mass of the cathode increased by 0.36g.Determine the R.A.M...
(Solved)
A current of 2.5A was passed through a cell containing N2+ ions for 25 minutes. The mass of the cathode increased by 0.36g.Determine the R.A.M of N. (F = 9.65 x 104Cmolˉ¹).
Date posted:
March 28, 2019
.
Answers (1)
-
The diagram below represents a set-up of apparatus used to investigate the effect of electric current on lead (II) bromide.
(Solved)
The diagram below represents a set-up of apparatus used to investigate the effect of electric current on lead (II) bromide.
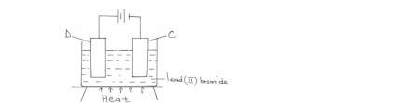
Describe what is observed at electrode C.
Date posted:
March 28, 2019
.
Answers (1)
-
25.0cm3 of 0.12M potassium hydroxide solution required 30.0cm3 of a solution of a dibasic acid (H2Y) for complete neutralization. The acid contained 3.15g per 500cm3...
(Solved)
25.0cm3 of 0.12M potassium hydroxide solution required 30.0cm3 of a solution of a dibasic acid (H2Y) for complete neutralization. The acid contained 3.15g per 500cm3 solution.
Calculate:
(a) The molarity of the acid solution
(b) The relative formula mass of the acid.
Date posted:
March 28, 2019
.
Answers (1)
-
The table below contains information regarding a species of helium.
(Solved)
The table below contains information regarding a species of helium.

Complete the table by indicating the numbers of electrons and neutrons.
Date posted:
March 28, 2019
.
Answers (1)
-
A Student in form four placed a thermometer in molten naphthalene at 850C and recorded the temperature and time until the naphthalene solidified. From the...
(Solved)
A Student in form four placed a thermometer in molten naphthalene at 850C and recorded the temperature and time until the naphthalene solidified. From the values obtained, the figure below was drawn.
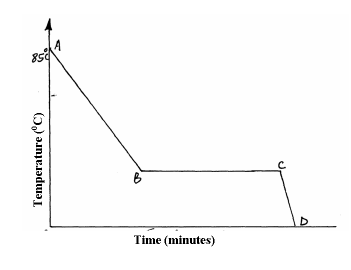
(a) What name is given to such a figure?
(b) Which part of the figure represents the change of state of naphthalene?
(c)In terms of kinetic theory. Explain what happens to molecules along AB.
Date posted:
March 28, 2019
.
Answers (1)
-
The table below shows information about three solid substances A, B and C. Study it and answer the question that follow.
Describe how you will separate...
(Solved)
The table below shows information about three solid substances A, B and C. Study it and answer the question that follow.
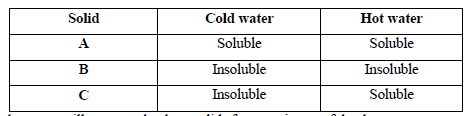
Describe how you will separate the three solids from a mixture of the three.
Date posted:
March 28, 2019
.
Answers (1)
-
Study the diagram below hence answer the questions that follow.
(Solved)
Study the diagram below hence answer the questions that follow.
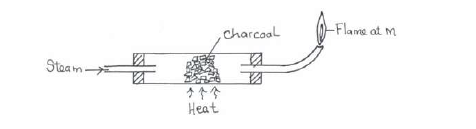
(i) Explain why it is necessary to have the flame at M.
(ii) Write down the equation for the reaction inside the apparatus.
Date posted:
March 28, 2019
.
Answers (1)
-
Calculate the volume of carbon (IV) oxide that would be produced if 15g of calcium carbonate reacted with 100cm³ of 2.0M hydrochloric acid (C =...
(Solved)
Calculate the volume of carbon (IV) oxide that would be produced if 15g of calcium carbonate reacted with 100cm³ of 2.0M hydrochloric acid (C = 12.0, O = 16.0, Ca = 40.0) molar gas volume = 24000cm³
Date posted:
March 28, 2019
.
Answers (1)
-
When lead (II) Carbonate is reacted with dilute sulphuric (VI) acid, the reaction takes place for a short time and then stops. Explain.
(Solved)
When lead (II) Carbonate is reacted with dilute sulphuric (VI) acid, the reaction takes place for a short time and then stops. Explain.
Date posted:
March 28, 2019
.
Answers (1)
-
Name the particles responsible for the electrical conductivity of:
(a) Graphite
(b) Magnesium Sulphate solution
(Solved)
Name the particles responsible for the electrical conductivity of:
(a) Graphite
(b) Magnesium Sulphate solution
Date posted:
March 28, 2019
.
Answers (1)
-
The diagram below illustrates how lithium would react with steam. Study it then answer the questions that follow.
(Solved)
The diagram below illustrates how lithium would react with steam. Study it then answer the questions that follow.

(a) Write the equation for the reaction that takes place.
(b) Explain why this experiment cannot be carried out with potassium in the same way as shown.
Date posted:
March 28, 2019
.
Answers (1)
-
The melting point of phosphorous (III) chloride is -910C while that of magnesium chloride is +7150C. In terms of structure and bonding explain the difference.
(Solved)
The melting point of phosphorous (III) chloride is -910C while that of magnesium chloride is +7150C. In terms of structure and bonding explain the difference.
Date posted:
March 28, 2019
.
Answers (1)
-
The grid below is part of the periodic table. Study it and answer the questions that follow. The letters are not actual symbols of elements.
(Solved)
The grid below is part of the periodic table. Study it and answer the questions that follow. The letters are not actual symbols of elements.
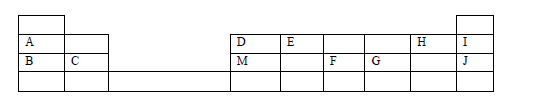
(a) What is the name given to the chemical family of element C?
(b) Would element B react with J? Explain.
(c) Compare the melting points of B and M.
Date posted:
March 28, 2019
.
Answers (1)
-
It is advisable to leave your flame in the luminous state when not in use. Give a reason why.
(Solved)
It is advisable to leave your flame in the luminous state when not in use. Give a reason why.
Date posted:
March 28, 2019
.
Answers (1)
-
The diagram below shows a non-luminous flame. Use it to answer the questions that follow:
(Solved)
The diagram below shows a non-luminous flame. Use it to answer the questions that follow:
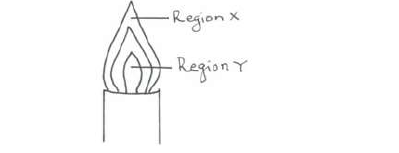
Two wooden splints were placed across regions X and Y respectively. Draw labelled diagrams to show the effects observed on the wooden splint placed across each region.
(i) Region X.
(ii) Region Y.
Date posted:
March 28, 2019
.
Answers (1)
-
The table below shows PH values of solutions A, B, C and D.
(Solved)
The table below shows PH values of solutions A, B, C and D.

(a) Identify a solution which is
(i) Strongly acidic. _________________________________________
(ii) Strongly basic _________________________________________
(b) Which to solutions would react with lead (II) oxide? Explain.
Date posted:
March 28, 2019
.
Answers (1)
-
Write the equation for decomposition of:
(a) Sodium nitrate.
(b) Copper (II) nitrate.
(Solved)
Write the equation for decomposition of:
(a) Sodium nitrate.
(b) Copper (II) nitrate.
Date posted:
March 28, 2019
.
Answers (1)