-
The set-up was used to prepare carbon (IV) oxide and investigate its properties.
(Solved)
The set-up was used to prepare carbon (IV) oxide and investigate its properties.

(a) Write an equation for the reaction in vessel B.
(b) What is the role of sodium hydroxide solution in the set-up?
(c) What would be observed if a burning splint is introduced at jet C?
Date posted:
March 29, 2019
.
Answers (1)
-
20cm³ of a dibasic acid required 25cm³ of 0.1M NaOH for complete neutralization.
(a) How many moles of sodium hydroxide reacted with the dibasic acid?
(b)...
(Solved)
20cm³ of a dibasic acid required 25cm³ of 0.1M NaOH for complete neutralization.
(a) How many moles of sodium hydroxide reacted with the dibasic acid?
(b) Calculate the concentration of the dibasic acid in moles per litre.
Date posted:
March 29, 2019
.
Answers (1)
-
0.92g of ethanol were found to burn in excess air producing a temperature rise of 32.5oC in 200cm3 of water(C=12.0,H=1.0,O=16.0) Density of water is 1g/cm3,...
(Solved)
0.92g of ethanol were found to burn in excess air producing a temperature rise of 32.5oC in 200cm3 of water(C=12.0,H=1.0,O=16.0) Density of water is 1g/cm3, specific heat capacity of water is 4.2KjKg-1K-1.
(a) Write the equation for the combustion of ethanol.
(b) Determine the molar heat of combustion of ethanol
Date posted:
March 29, 2019
.
Answers (1)
-
The diagram below shows electrolysis of dilute copper (II) sulphate solution using copper electrodes;
(Solved)
The diagram below shows electrolysis of dilute copper (II) sulphate solution using copper electrodes;

a) State the observations made at electrode A and B
b) Write the equation of reaction at electrode A
Date posted:
March 29, 2019
.
Answers (1)
-
Below is a simplified process for manufacture of sodium carbonate by Solvay process.
a) Name substance M.
b) Name the process taking place in step II.
c)...
(Solved)
Below is a simplified process for manufacture of sodium carbonate by Solvay process.

a) Name substance M.
b) Name the process taking place in step II.
c) Write an equation for the reaction in step III.
d) What is the fate of ammonium chloride in the solvay process.
Date posted:
March 29, 2019
.
Answers (1)
-
Using dots (.) and crosses (x) to represent electrons, show the bonding in hydroxonium ion (H=1 O=8).
(Solved)
Using dots (.) and crosses (x) to represent electrons, show the bonding in hydroxonium ion (H=1 O=8).
Date posted:
March 29, 2019
.
Answers (1)
-
In an experiment, two pieces of iron sheets were wrapped in each case with zinc and copper metal sheets as shown below.They were left in...
(Solved)
In an experiment, two pieces of iron sheets were wrapped in each case with zinc and copper metal sheets as shown below.They were left in the open for some months.

State and explain the observations made in the experiments; (I), (II)
Date posted:
March 28, 2019
.
Answers (1)
-
Name the following processes;
a) When anhydrous calcium chloride is left in an open beaker overnight a solution was formed.
b) When sodium carbonate decahydrate crystals...
(Solved)
Name the following processes;
a) When anhydrous calcium chloride is left in an open beaker overnight a solution was formed.
b) When sodium carbonate decahydrate crystals are left in an open beaker for some days it turned into a powder.
Date posted:
March 28, 2019
.
Answers (1)
-
60cm³ of oxygen gas diffused through a porous hole in 50 seconds. How long will it take 80cm³ of sulphur (IV) oxide to diffuse through...
(Solved)
60cm³ of oxygen gas diffused through a porous hole in 50 seconds. How long will it take 80cm³ of sulphur (IV) oxide to diffuse through the same hole under the same conditions (S = 32, O = 16).
Date posted:
March 28, 2019
.
Answers (1)
-
Starting with solid lead (II) carbonate, briefly describe how a sample of lead (II) chloride can be prepared.
(Solved)
Starting with solid lead (II) carbonate, briefly describe how a sample of lead (II) chloride can be prepared.
Date posted:
March 28, 2019
.
Answers (1)
-
a) Explain why hydrogen gas has been replaced by helium in filling aeroplane tyres.
b)Hydrogen gas is bubbled through oil in presence of a catalyst to...
(Solved)
a) Explain why hydrogen gas has been replaced by helium in filling aeroplane tyres.
b)Hydrogen gas is bubbled through oil in presence of a catalyst to yield fats.
(i) Name a suitable catalyst used in the above process.
(ii) What name is given to the above process.
Date posted:
March 28, 2019
.
Answers (1)
-
4. 9 g a tribasic acid was dissolved in water and the solution made up to 500cm3. If the molarity of the hydrogen ions in...
(Solved)
4. 9 g a tribasic acid was dissolved in water and the solution made up to 500cm3. If the molarity of the hydrogen ions in the solution is 0.3 molar calculate the relative molecular mass of the acid.
Date posted:
March 28, 2019
.
Answers (1)
-
The diagram below represents pipes used in Frasch pump for the extraction of sulpur
Name the substance that pass through the following tubes and the direction...
(Solved)
The diagram below represents pipes used in Frasch pump for the extraction of sulpur

Name the substance that pass through the following tubes and the direction which they flow.
1.
2.
3.
Date posted:
March 28, 2019
.
Answers (1)
-
12.0 cm3 of methane and 48cm3 of oxygen were exploded together. The final volume measured under the original conditions was 36.0 cm3 neglecting the water...
(Solved)
12.0 cm3 of methane and 48cm3 of oxygen were exploded together. The final volume measured under the original conditions was 36.0 cm3 neglecting the water formed. 24.0cm3 of this was unused oxygen. Show the ratio of reacting volume of the gases referred to and gaseous products formed.
Date posted:
March 28, 2019
.
Answers (1)
-
Element X contains isotopes with mass number 16 and 18 respectively existing in the ratio
1: 3. Calculate the relative atomic mass of X.
(Solved)
Element X contains isotopes with mass number 16 and 18 respectively existing in the ratio
1: 3. Calculate the relative atomic mass of X.
Date posted:
March 28, 2019
.
Answers (1)
-
What volume of methane would remain if a burner containing 40cm³ of methane burns in 40cm³ of enclosed air
(assuming that oxygen is 20% of air)?
(Solved)
What volume of methane would remain if a burner containing 40cm³ of methane burns in 40cm³ of enclosed air
(assuming that oxygen is 20% of air)?
Date posted:
March 28, 2019
.
Answers (1)
-
A compound has empirical formula C3H6O and relative formula mass of 116. Determine it’s molecular formula.(C = 12, H = 1, O = 16).
(Solved)
A compound has empirical formula C3H6O and relative formula mass of 116. Determine it’s molecular formula.(C = 12, H = 1, O = 16).
Date posted:
March 28, 2019
.
Answers (1)
-
The scheme below shows some reaction of salt. Study it and answer questions that follow.
(Solved)
The scheme below shows some reaction of salt. Study it and answer questions that follow.
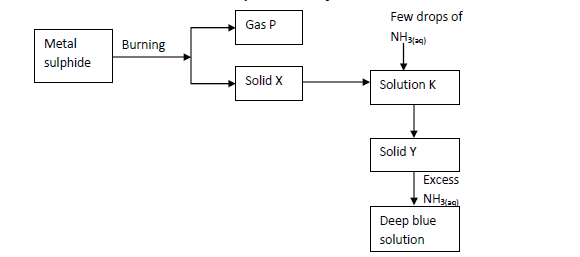
(i) Write an equation for the reaction to show formation of gas P and solid X.
(ii) Give the name and formula of the complex ion responsible for the deep blue colour in the solution.
Date posted:
March 28, 2019
.
Answers (1)
-
Explain why potassium chloride conducts electricity in both the molten state and in aqueous solution whereas hydrogen chloride conducts electricity only in aqueous solution and...
(Solved)
Explain why potassium chloride conducts electricity in both the molten state and in aqueous solution whereas hydrogen chloride conducts electricity only in aqueous solution and not in gaseous state.
Date posted:
March 28, 2019
.
Answers (1)
-
An element E has relative atomic mass of 69.39. Given that the element has two isotopes of atomic masses 62.35 and 70.45, calculate the relative...
(Solved)
An element E has relative atomic mass of 69.39. Given that the element has two isotopes of atomic masses 62.35 and 70.45, calculate the relative abundance of each of the isotopes.
Date posted:
March 28, 2019
.
Answers (1)