In aqueous solutions there are usually four ions present, two from the water and two from
the dissolved salt. The products will be gaseous, or metals which are deposited on the
negative electrode. It is composed of an open cylinder of glass approximately 8 cm high and
2.5 cm in diameter. A small bottle of similar size with the bottom cut off would do just as
well. The cylinder has a 2-hole rubber stopper carrying two carbon electrodes with
connecting leads to a battery, or DC supply of 4 to 6 volts. If cork is used, this must be
made leak proof by covering the whole of the bottom surface round the electrodes and the
glass edge with Faraday's wax or a similar soft wax. The electrodes may be carbon rods
from a dry cell or pencil leads. The alloy supports for the coiled filament in electric light
bulbs have also been found suitable for electrodes. The electrodes should project about 2
cm into the cylinder and also 2 cm below for attaching the leads to the battery. Pencil leads
are brittle, and if they are used it is better to fix the electrodes in the following way.
Solder a piece of stout copper wire to a 4 cm length of braided copper screening wire. Drill
two holes in the rubber stopper with a 1 mm drill. Insert the copper wire into the hole from
above and pull it right through the stopper until the screening wire is also pulled a little way
into the hole. Into the core of this screening wire insert the pencil lead securely. Then pull
the screening wire with the lead further into the stopper so that the lead electrode is
firmly held in the stopper. The excess copper wire is cut off. This procedure is repeated
with the other electrode. The solution is placed in the glass cylinder. The two small tubes
are then filled with the solution, and carefully inverted over the electrodes. The electrodes
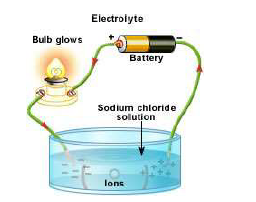
are connected to a safe DC supply with a small bulb in series. Increase the voltage until the
bulb lights, showing that a current is flowing. When this happens, cut out the bulb from the
circuit by closing the switch, as shown. This will allow a larger current to flow. The tubes
collect any gas given off and the properties of the gas should be tested. Using carbon
electrodes, the following results will be found.
sharon kalunda answered the question on
April 12, 2019 at 08:22