Candle goes off.
Carbon (IV) oxide produced turns lime water white// formation of a white
precipitate due to the reaction between CO2 and limewater.
sharon kalunda answered the question on May 17, 2019 at 08:29
- The diagram below outlines industrial preparation of bleaching powder.(Solved)
The diagram below outlines industrial preparation of bleaching powder.
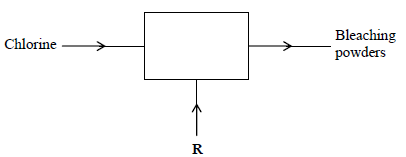
(i) Give the chemical name of bleaching powder
(ii) Identify substance R
(iii) Explain why water in which bleaching powder has been added needs a lot of soap
during washing.
Date posted: May 17, 2019. Answers (1)
- The reaction of water and calcium gave gas Q collected as in the diagram below(Solved)
The reaction of water and calcium gave gas Q collected as in the diagram below
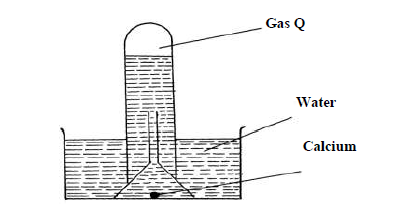
(i) Identify gas Q
(ii) Explain why the solution left after the reaction is a weak base.
Date posted: May 17, 2019. Answers (1)
- On heating Ammonium Chloride two gas P and J were evolved G turned moist litmus
paper red and J turned Moist litmus paper blue. On cooling...(Solved)
On heating Ammonium Chloride two gas P and J were evolved G turned moist litmus
paper red and J turned Moist litmus paper blue. On cooling , the two gases
recombined to form a white solid
a) Identify P and J
b) What property of Ammonium Chloride is shown in this experiment?
Date posted: May 17, 2019. Answers (1)
- A solution of hydrogen chloride gases in water reacts with Zinc carbonate, but a solution of hydrogen chloride in methylbenzene does not . Explain(Solved)
A solution of hydrogen chloride gases in water reacts with Zinc carbonate, but a solution of hydrogen chloride in methylbenzene does not. Explain
Date posted: May 17, 2019. Answers (1)
- Dry hydrogen chloride gas was passed through heated iron wire as shown in the diagram
below(Solved)
Dry hydrogen chloride gas was passed through heated iron wire as shown in the diagram
below
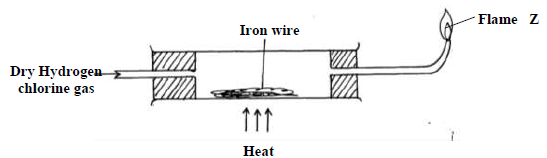
a) (i) How can the identity of the substance burning with flame Z be confirmed.
(ii) What is observed in combustion tube during the experiment?
(iii) Write the equation for the reaction taking place in the combustion tube.
(iv) Chlorine gas was passed over the product obtained in the combustion tube to give
another product Q
a) State one precaution that should be taken. Explain
b) Identify product Q
c) The total mass of product Q formed was found to be 5.3g. Calculate the volume of
chlorine gas used.
(Cl = 35.5, Fe= 56, Molar gas volume at room temperature = 2400cm3)
Date posted: May 17, 2019. Answers (1)
- The flow chart below outlines some of the process involved during extraction of copper.(Solved)
The flow chart below outlines some of the process involved during extraction of copper.

(i) Write the formula of copper pyrite.
(ii) Name liquid T
(iii) Write equations for the reactions taking place in the 2nd roasting furnace.
(iv) Identify substance B and write equation for the reaction that take place in the smelting furnace.
(v) State the purpose of substance F.
Date posted: May 17, 2019. Answers (1)
- Study the scheme below and answer the questions that follow.(Solved)
Study the scheme below and answer the questions that follow.

(i) Identify the product B.
(ii) Name the compounds C and E.
(iii) State the conditions for step 1
(iv) Write the equation for the reaction leading tot the formation of methane.
(v) State two industrial uses of methane.
(vi) Identify the reagent D
Date posted: May 17, 2019. Answers (1)
- Name the following compounds.(Solved)
Name the following compounds.
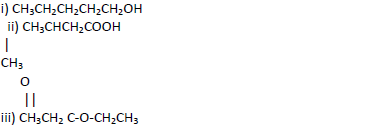
Date posted: May 17, 2019. Answers (1)
- The graph below was obtained from an experiment used to investigate the reaction
between Zinc granules and 2M hydrochloric acid.(Solved)
The graph below was obtained from an experiment used to investigate the reaction
between Zinc granules and 2M hydrochloric acid.
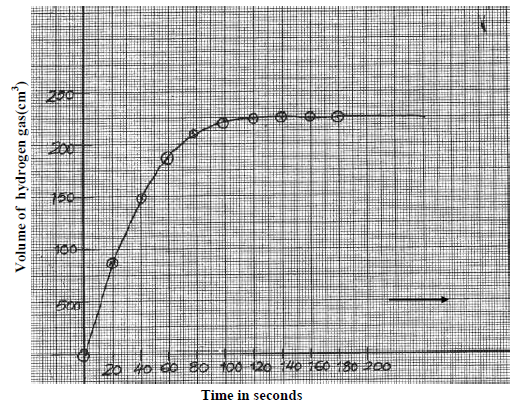
a) Calculate the rate of reaction when t = 60seconds.
b) Suggest how the rate of the above reaction can be reduced so that it can be studied more closely at the same temperature.
Date posted: May 17, 2019. Answers (1)
- Describe two observable characteristics of a luminous flame(Solved)
Describe two observable characteristics of a luminous flame.
Date posted: May 17, 2019. Answers (1)
- Write a chemical equation for the combustion of laboratory gas , when the
Bunsen burner produces a non-luminous flame.(Solved)
Write a chemical equation for the combustion of laboratory gas , when the
Bunsen burner produces a non-luminous flame.
Date posted: May 17, 2019. Answers (1)
- Explain why during the extraction of metals, copper can be extracted by the electrolysis of
copper (II) sulphate solution, while aluminium chloride cannot be extracted by...(Solved)
Explain why during the extraction of metals, copper can be extracted by the electrolysis of
copper (II) sulphate solution, while aluminium chloride cannot be extracted by electrolysis of
aluminium sulphate solution.
Date posted: May 17, 2019. Answers (1)
- Study the following equilibrium equation and answer the questions that follows.(Solved)
Study the following equilibrium equation and answer the questions that follows.
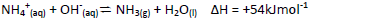
Explain how ammonia gas can be prepared in the laboratory.
Date posted: May 17, 2019. Answers (1)
- The following are standard electrode potentials of some half-cell reactions. Use the data to
answer the questions that follow.(Solved)
The following are standard electrode potentials of some half-cell reactions. Use the data to
answer the questions that follow.
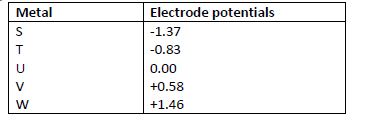
i) Suggest the identify of element U.
ii) Draw a labeled diagram of an electrochemical cell that would produce the largest e.m.f.
Date posted: May 17, 2019. Answers (1)
- The following diagram represents a set-up used to investigate conditions necessary for rusting of iron.(Solved)
The following diagram represents a set-up used to investigate conditions necessary for rusting of iron.
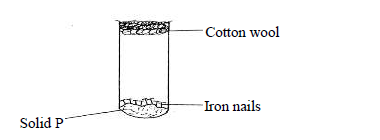
After several days it was found that the nails did not rust. Identify solid P.
Date posted: May 17, 2019. Answers (1)
- The following diagram was used to investigate the electrolysis of copper(II) Sulphate solution
and molten G chloride using carbon electrodes.(Solved)
The following diagram was used to investigate the electrolysis of copper(II) Sulphate solution
and molten G chloride using carbon electrodes.
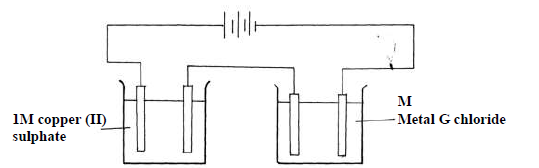
When a fixed current was passed through the two electrolytic cells as shown, 1.27g of copper
and 0.6g of G were deposited at the respective electrodes,. Calculate the numerical value
of x in the formula Gx+
(Cu=63.5; g = 60; 1 Faraday = 96500C)
Date posted: May 17, 2019. Answers (1)
- In the chemistry laboratory, both blue and red litmus papers are used to test for the nature
of gases and solutions. Explain(Solved)
In the chemistry laboratory, both blue and red litmus papers are used to test for the nature
of gases and solutions. Explain
Date posted: May 17, 2019. Answers (1)
- Explain why graphite is preferred to lubricating oil in the moving parts of the machine.(Solved)
Explain why graphite is preferred to lubricating oil in the moving parts of the machine.
Date posted: May 17, 2019. Answers (1)
- Q grams of a radioactive isotope sample takes 80 days to disintegrate to 7g. The halflife of the isotope is 20days. Find the initial mass...(Solved)
Q grams of a radioactive isotope sample takes 80 days to disintegrate to 7g. The halflife of the isotope is 20days. Find the initial mass Q.
Date posted: May 17, 2019. Answers (1)
- In the laboratory, chlorine gas can be prepared by the reaction;(Solved)
In the laboratory, chlorine gas can be prepared by the reaction;

Given the following half- cell reactions;
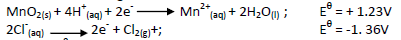
Use the Eθ cell to explain how the above reaction is carried out in the laboratory.
Date posted: May 17, 2019. Answers (1)