(a) 2,8,8
(b) 2,8,2
sharon kalunda answered the question on May 17, 2019 at 14:11
- Study the reduction potentials below.
(i) Identify the weakest oxidizing agent.
(ii) Calculate the e.m.f of the cell that would produce the highest output of voltage.
(iii)...(Solved)
Study the reduction potentials below.

(i) Identify the weakest oxidizing agent.
(ii) Calculate the e.m.f of the cell that would produce the highest output of voltage.
(iii) Write the cell diagram for the cell formed above.
Date posted: May 17, 2019. Answers (1)
- Study the diagram below and answer the questions which follow.(Solved)
Study the diagram below and answer the questions which follow.
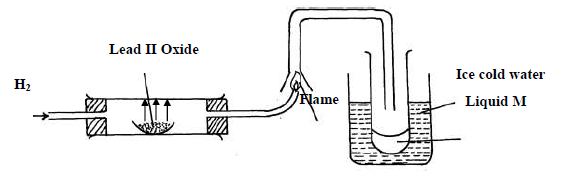
(i) State two observations made when hydrogen gas pass over hot lead II oxide.
(ii) Write the equation for the reaction which occurs in the combustion tube.
(iii) What property of hydrogen is shown in the experiment above
(iv) Identify liquid M.
(v) What type of reaction occurs when hydrogen gas reacts with butene?
(vi) State the condition required for the reaction (v) above
(vii) Apart from hydrogen peroxide, state two other reagents that can be used to prepare
oxygen gas.
(viii) Write an equation to show how hydrogen gas is formed from the reagents chosen in
(vii) above.
Date posted: May 17, 2019. Answers (1)
- What is atomicity?(Solved)
What is atomicity?
Date posted: May 17, 2019. Answers (1)
- Study the periodic grid below and answer the questions which follow. The letters do not
represent actual symbols of the elements.(Solved)
Study the periodic grid below and answer the questions which follow. The letters do not
represent actual symbols of the elements.
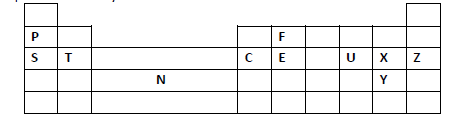
(i) To which category of elements does element N belong?
(ii) Compare the atomic radius of element U and X. Explain.
(iii) An ion A3- has a configuration of 2.8. Place element A on the grid above.
(iv) Which of the group 1 elements will require the greatest amount of energy to
remove the outermost electron. Explain.
(v) Why is element Z used in light bulbs?
(vi) Write the formula of the phosphate of element T.
(vii) State the type of structure found in the oxide of element F.
Date posted: May 17, 2019. Answers (1)
- A given amount of propane was used to heat one litre of water. The temperature of
the water rose from 25oC to 50.5oC. (S.H.C of water...(Solved)
A given amount of propane was used to heat one litre of water. The temperature of
the water rose from 25oC to 50.5oC. (S.H.C of water = 4.2J/g/k)
(i) Calculate the heat change for the reaction.
(ii) Find the mass of propane burnt (C=12, H=1)
Date posted: May 17, 2019. Answers (1)
- Study the heats of combustion shown below.(Solved)
Study the heats of combustion shown below.

Draw an energy cycle diagram linking heat of formation of propane with its heat of
combustion and the heat of combustion of the constituent elements.
Date posted: May 17, 2019. Answers (1)
- Define standard heat of combustion of a substance.(Solved)
Define standard heat of combustion of a substance.
Date posted: May 17, 2019. Answers (1)
- Study the diagram below. State and explain the observation made after sometime(Solved)
Study the diagram below.

State and explain the observation made after sometime.
Date posted: May 17, 2019. Answers (1)
- Give an equation to show how chlorine forms bleaching powder.(Solved)
Give an equation to show how chlorine forms bleaching powder.
Date posted: May 17, 2019. Answers (1)
- Chlorine reacts with cold dilute sodium hydroxide to form a bleaching agent.
Name the bleaching agent.(Solved)
Chlorine reacts with cold dilute sodium hydroxide to form a bleaching agent.
Name the bleaching agent.
Date posted: May 17, 2019. Answers (1)
- The set up below was used to prepare chlorine gas. (i) Identify solid M (ii) What is the role of water in the experiment?.....(Solved)
The set up below was used to prepare chlorine gas.

(i) Identify solid M
(ii) What is the role of water in the experiment?
(iii) Complete the set up to show how dry chlorine gas can be collected
(iv) Write a chemical equation to show how chlorine gas is formed.
Date posted: May 17, 2019. Answers (1)
- State one use of lead.(Solved)
State one use of lead.
Date posted: May 17, 2019. Answers (1)
- Pure lead can be obtained by electrolysis. Identify the anode and cathode for the
process.(Solved)
Pure lead can be obtained by electrolysis. Identify the anode and cathode for the
process.
Date posted: May 17, 2019. Answers (1)
- The flow chart below summarizes the process of extraction of lead from a chief ore.(Solved)
The flow chart below summarizes the process of extraction of lead from a chief ore.

(i) Identify process T
(ii) Give the name of:
Gas N
Solid F
(iii) Give two functions of CaCO3 in the extraction process.
(iv) Write an equation to show how waste product J is formed.
Date posted: May 17, 2019. Answers (1)
- Name any two ores of lead.(Solved)
Name any two ores of lead.
Date posted: May 17, 2019. Answers (1)
- a)Write down the electron arrangement for an atom of element U which has a mass number 14 and
contains 8 neutrons.(Solved)
a)Write down the electron arrangement for an atom of element U which has a mass number 14 and
contains 8 neutrons.
(b) Draw the structure of an atom of A given in (a) above.
Date posted: May 17, 2019. Answers (1)
- A sealed glass tube containing 250 cm3 of nitrogen gas at r.t.p was immersed in boiling water. Calculate the pressure inside the tube if the...(Solved)
A sealed glass tube containing 250 cm3 of nitrogen gas at r.t.p was immersed in boiling water. Calculate the pressure inside the tube if the volume of the gas does not change due to expansion of glass. (Room pressure=760mmHg, room temperature=298K).
Date posted: May 17, 2019. Answers (1)
- Explain this observation:
When hydrogen chloride gas is dissolved in water, the solution conducts electricity while a
solution of hydrogen chloride gas in propanone does not conduct...(Solved)
Explain this observation:
When hydrogen chloride gas is dissolved in water, the solution conducts electricity while a
solution of hydrogen chloride gas in propanone does not conduct electricity
Date posted: May 17, 2019. Answers (1)
- A solution of bromine in methyl benzene turns colourless when butane gas is passed through it.(Solved)
A solution of bromine in methyl benzene turns colourless when butane gas is passed through it.
(a) What type of reaction takes place?
(b) Write equation of the reaction which takes place.
Date posted: May 17, 2019. Answers (1)
- The thermochemical equation below shows a dynamic equilibrium between hydrogen iodide
gas and its elements:(Solved)
The thermochemical equation below shows a dynamic equilibrium between hydrogen iodide
gas and its elements:
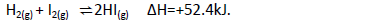
(a) Explain how the following changes would affect the production of hydrogen iodide.
(i) increase in temperature
(ii) decrease in pressure.
(b) Calculate the molar enthalpy for formation of HI (g).
Date posted: May 17, 2019. Answers (1)