(i) P and S; ionic radii larger than their atomic radii II gain of electrons causes increase in ionic radius;
(ii) Oxide of R has strong ionic bonds with giants ionic structure; while oxide of S has weak vander waals forces with simple molecular structure;
(iii) P and Q; P is a non-metal with the smaller atomic radius hence most electronegative;Q is a metal with the largest atomic radius hence most electropositive;
sharon kalunda answered the question on May 21, 2019 at 12:15
- What is the difference between thermosoftening and thermosetting plastics?(Solved)
What is the difference between thermosoftening and thermosetting plastics?
Date posted: May 21, 2019. Answers (1)
- Draw a dot (.) and cross (x) diagram to show bonding in:-
(i) Ammonium ion (NH4)
(ii) Silane (SiH4)
(N=14...(Solved)
Draw a dot (.) and cross (x) diagram to show bonding in:-
(i) Ammonium ion (NH4)
(ii) Silane (SiH4)
(N=14 H=1 Si=14)
Date posted: May 21, 2019. Answers (1)
- Lead (ii)nitrate was heated strongly for some time.
(i) State two observations made during heating.
(ii) Write an equation for the reaction.(Solved)
Lead (ii)nitrate was heated strongly for some time.
(i) State two observations made during heating.
(ii) Write an equation for the reaction.
Date posted: May 21, 2019. Answers (1)
- Consider the following electrochemical cell.
(i) Name the electrodes for the above cell.
(ii) Write the electrodes for the above cell
(iii) Name a possible salt...(Solved)
Consider the following electrochemical cell.

(i) Name the electrodes for the above cell.
(ii) Write the electrodes for the above cell
(iii) Name a possible salt bridge.
Date posted: May 21, 2019. Answers (1)
- A volume of nitrogen gas diffuses through a porous pot in 70 seconds. How long would it take
400cm3 of carbon (iv) oxide to diffuse through...(Solved)
A volume of nitrogen gas diffuses through a porous pot in 70 seconds. How long would it take
400cm3 of carbon (iv) oxide to diffuse through the same porous pot? (C=12 O=16 N=14)
Date posted: May 21, 2019. Answers (1)
- When a few drops of aqueous ammonia were added to copper (ii) chloride solution, a light blue
precipitate was formed. On addition of excess ammonia solution,...(Solved)
When a few drops of aqueous ammonia were added to copper (ii) chloride solution, a light blue
precipitate was formed. On addition of excess ammonia solution, a deep blue solution was formed.
(a) Identify the substance responsible for the:-
(i) light blue precipitate.
(ii) deep blue solution.
(b) Write an equation for the reaction leading to observation in (a) (ii) above.
Date posted: May 21, 2019. Answers (1)
- Pieces of blue and red litmus papers were placed into a beaker containing water into which
Aluminium Chloride had been dissolved.State the observations made on the...(Solved)
Pieces of blue and red litmus papers were placed into a beaker containing water into which
Aluminium Chloride had been dissolved.State the observations made on the papers. Explain your answer.
Date posted: May 21, 2019. Answers (1)
- Is dissolving of aluminium chloride in water a physical or chemical process? Explain(Solved)
Is dissolving of aluminium chloride in water a physical or chemical process? Explain
Date posted: May 21, 2019. Answers (1)
- During electrolysis of copper (ii) sulphate solution using graphite electrodes, a current of 2
amperes was passed for 15 minutes. Determine the mass of the products...(Solved)
During electrolysis of copper (ii) sulphate solution using graphite electrodes, a current of 2
amperes was passed for 15 minutes. Determine the mass of the products at the cathode.
(1F=96,500C Cu=63.5)
Date posted: May 21, 2019. Answers (1)
- Explain why graphite conducts electricity while diamond does not.(Solved)
Explain why graphite conducts electricity while diamond does not.
Date posted: May 21, 2019. Answers (1)
- The following pairs of compounds were reacted together and the maximum temperature rise
recorded for each reaction.
A- 50 cm3 of 2M ammonia solution and 50 cm3...(Solved)
The following pairs of compounds were reacted together and the maximum temperature rise
recorded for each reaction.
A- 50 cm3 of 2M ammonia solution and 50 cm3 of 2M ethanoic acid.
B- 50 cm3 of 2M sodium hydroxide and 50 cm3 of 2M hydrochloric acid.
C- 50 cm3 of 2M sodium hydroxide and 50 cm3 of 2M ethanoic acid.
(a) State the pair which showed:-
(i) the highest temperature rise.
(ii) the lowest temperature rise.
(b) Explain your answers above.
Date posted: May 21, 2019. Answers (1)
- Radium 226, whose atomic number is 88, undergoes beta decay to form a new element X.Write an equation for this change.(Solved)
Radium 226, whose atomic number is 88, undergoes beta decay to form a new element X.Write an equation for this change.
Date posted: May 21, 2019. Answers (1)
- The table below shows the number of valence electrons in elements D, E and F.
(i) Explain why D and E would not be expected to...(Solved)
The table below shows the number of valence electrons in elements D, E and F.
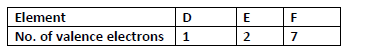
(i) Explain why D and E would not be expected to react together to form a compound.
(ii) Write a chemical equation to show the effect of heat on a carbonate of E.
Date posted: May 21, 2019. Answers (1)
- The structure below shows the repeat unit showed in a polymer.
(i) Name the polymer.
(ii) Draw the structures of the two monomers forming the polymer.(Solved)
The structure below shows the repeat unit showed in a polymer.
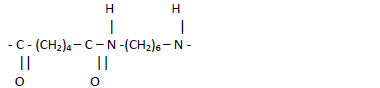
(i) Name the polymer.
(ii) Draw the structures of the two monomers forming the polymer.
Date posted: May 21, 2019. Answers (1)
- Name the solution and the catalyst used in preparation of oxygen in the laboratory.(Solved)
Name the solution and the catalyst used in preparation of oxygen in the laboratory.
Date posted: May 21, 2019. Answers (1)
- When 31.2g of hydrated. Aluminium oxide ( Al2O3XH2O) was heated to a constant mass of 20.6g of Aluminium oxide ( Al2O3) was obtained. Determine the value...(Solved)
When 31.2g of hydrated. Aluminium oxide ( Al2O3XH2O) was heated to a constant
mass of 20.6g of Aluminium oxide ( Al2O3) was obtained. Determine the value of x in
hydrated oxide.(Al= 27.0, O=16.0, H=1.0)
Date posted: May 21, 2019. Answers (1)
- Aluminium is not used for marine purpose.Give a reason.(Solved)
Aluminium is not used for marine purpose.Give a reason.
Date posted: May 21, 2019. Answers (1)
- Carbon is not used for the reduction of Aluminium oxides.Give a reason(Solved)
Carbon is not used for the reduction of Aluminium oxides.Give a reason
Date posted: May 21, 2019. Answers (1)
- The flow chart below shows industrial extraction Aluminium metal. Study it and answer the questions that follow.
(i) Explain how process T is carried out.
(ii)...(Solved)
The flow chart below shows industrial extraction Aluminium metal. Study it and answer the questions that follow.

(i) Explain how process T is carried out.
(ii) Name residue P, give a reason.
(iii) Explain why it is necessary to heat Aluminium oxide in presence of cryolite before
electrolysis is carried out.
Date posted: May 21, 2019. Answers (1)
- Describe how sulphuric acid is manufactured from sulphur (VI) oxide.(Solved)
Describe how sulphuric acid is manufactured from sulphur (VI) oxide.
Date posted: May 21, 2019. Answers (1)