- Study the information in the table below and answer the questions that follow.
(i) Write the general formula of the hydrocarbons in the table.
(ii) Predict...(Solved)
Study the information in the table below and answer the questions that follow.
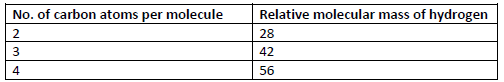
(i) Write the general formula of the hydrocarbons in the table.
(ii) Predict the relative molecular mass of the hydrocarbon with 5 carbon atoms.
(iii) Determine the molecular formula of the hydrocarbon in (ii) and draw its structural
formula.
Date posted: May 22, 2019. Answers (1)
- Describe how the mixture of Ammonium chloride, sodium chloride and lead II chloride can be separated if all the components of the mixture are to...(Solved)
Describe how the mixture of Ammonium chloride, sodium chloride and lead II chloride can be separated if all the components of the mixture are to be recovered.
Date posted: May 22, 2019. Answers (1)
- The diagram below represents an arrangement for a large scale manufacture of ethanol for domestic consumption.
(i) Name the process by which ethanol is obtained from...(Solved)
The diagram below represents an arrangement for a large scale manufacture of ethanol for domestic consumption.
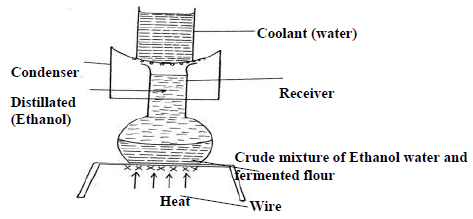
(i) Name the process by which ethanol is obtained from the crude oil.
(ii) Suggest two reasons why water is a coolant in this process.
(iii) Why is it possible to separate ethanol from the mixture by this process.
Date posted: May 22, 2019. Answers (1)
- Study the energy level below and answer the questions that follow.
(i) State and explain whether the reaction represented in the diagram is endothermic or
exothermic.
(ii)...(Solved)
Study the energy level below and answer the questions that follow.
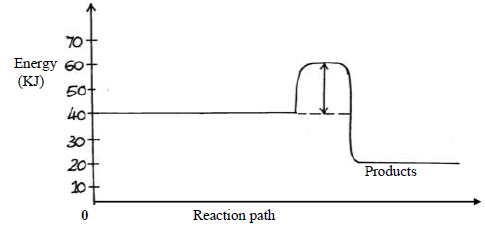
(i) State and explain whether the reaction represented in the diagram is endothermic or
exothermic.
(ii) From the diagram, determine; the activation energy
Date posted: May 22, 2019. Answers (1)
- Consider the reaction represented by the equation:
Explain the effect of the following on the reaction;
(a) An increase in pressure
(b) Increase in temperature(Solved)
Consider the reaction represented by the equation:
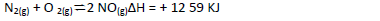
Explain the effect of the following on the reaction;
(a) An increase in pressure
(b) Increase in temperature
Date posted: May 22, 2019. Answers (1)
- In an experiment, a few drops of concentrated nitric (IV)acid were added to aqueous Iron (II)
sulphate in a test tube. excess sodium hydroxide solution was...(Solved)
In an experiment, a few drops of concentrated nitric (IV)acid were added to aqueous Iron (II)
sulphate in a test tube. excess sodium hydroxide solution was then added to the mixture.
(a) State the observations that were made when:
(i) Concentrated nitric (V) acid was added to aqueous Iron (II) sulphate.
(ii) Excess sodium hydroxide was added to the mixture.
(b) Write an ionic equation for the reaction that occurred in a(ii) above.
Date posted: May 22, 2019. Answers (1)
- The set up below was used to investigate a chemical property of carbon. Study it and answer
the questions that follow.
(i) What observations were made on...(Solved)
The set up below was used to investigate a chemical property of carbon. Study it and answer
the questions that follow.
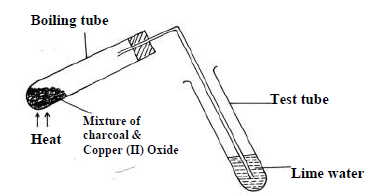
(i) What observations were made on heating the mixture.
(ii) What is the industrial application of carbon in terms of property investigated
above.
Date posted: May 22, 2019. Answers (1)
- Identify the type of bond formed in (i) and (ii) .(Solved)
Identify the type of bond formed in (i) and (ii) .
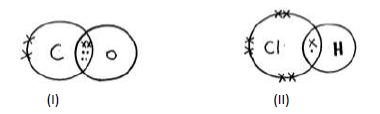
Date posted: May 22, 2019. Answers (1)
- Zinc carbonate decomposes on heating producing a gaseous product and a residue. What
volume of the gaseous product at s.t.p is produced from 2.5 g of...(Solved)
Zinc carbonate decomposes on heating producing a gaseous product and a residue. What
volume of the gaseous product at s.t.p is produced from 2.5 g of the carbonate? ( Zn = 65,
C=12,O=16 M.G.V at s.t.p = 22400cm3)
Date posted: May 22, 2019. Answers (1)
- If aqueous lead (II) nitrate is added to aqueous solution potassium iodide, a bright yellow
precipitate is formed.
(i) Write down the formula of the precipitate formed.
(ii)...(Solved)
If aqueous lead (II) nitrate is added to aqueous solution potassium iodide, a bright yellow
precipitate is formed.
(i) Write down the formula of the precipitate formed.
(ii) Write an ionic equation for the reaction above.
Date posted: May 22, 2019. Answers (1)
- Study the information in the table below and answer the questions that follow. The letters
do not represent the actual symbols of the elements.(Solved)
Study the information in the table below and answer the questions that follow. The letters
do not represent the actual symbols of the elements.

(i) Write the formulae of carbonate R and M
(ii) Describe how the carbonate of M can be obtained from a mixture of carbonate R and
M.
(iii) R is more reactive than L. Explain
Date posted: May 22, 2019. Answers (1)
- The diagram below shows a set-up of apparatus used to separate miscible liquids.
(a) Name the parts labelled A and B
(b) State the function of...(Solved)
The diagram below shows a set-up of apparatus used to separate miscible liquids.
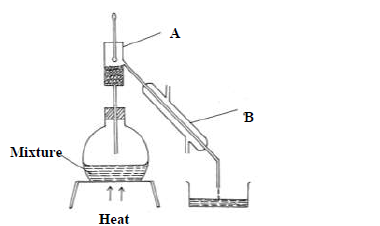
(a) Name the parts labelled A and B
(b) State the function of the part labeled A.
(c) State the property of the mixture that makes it suitable to be separated by the
method above.
Date posted: May 22, 2019. Answers (1)
- The table represents results on four samples of water. Study it an answer the
question that follows.
Which sample is likely to be temporary hard water? Explain(Solved)
The table represents results on four samples of water. Study it an answer the
question that follows.
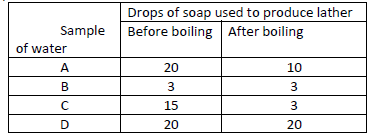
Which sample is likely to be temporary hard water? Explain
Date posted: May 22, 2019. Answers (1)
- Use the flow chart below to answer the questions that follow
a) Name the following;
i) Gas L
ii) Gas H
iii) K
b) Name the processes...(Solved)
Use the flow chart below to answer the questions that follow
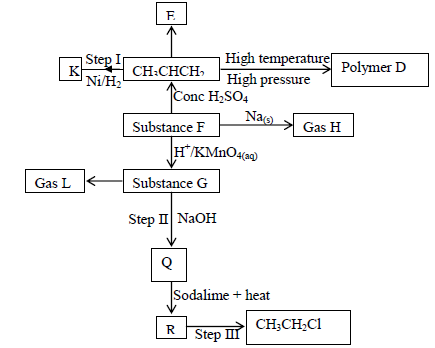
a) Name the following;
i) Gas L
ii) Gas H
iii) K
b) Name the processes involved in the following steps
i) Step I
ii) Step II
iii) Step III
c) Write a chemical equation for the complete combustion of substance F
d) Name the condition and reagents in step III
i) Condition
ii) Reagent
e) Calculate the mass of salt Q that would be formed by using 21.9kg of G when
it reacts with excess sodium hydroxide
(C = 120, H = 1.0, Na = 23.0, O = 16.0)
Date posted: May 22, 2019. Answers (1)
- Below is a list of potential differences obtained when metal P, Q, R, S and T are used
in the following electrochemical cell(Solved)
Below is a list of potential differences obtained when metal P, Q, R, S and T are used
in the following electrochemical cell
Metal (s) / Metal ions // Copper (II) ions / copper (s)
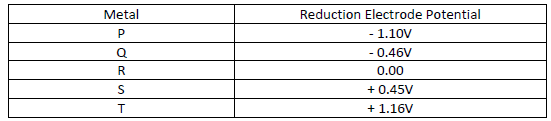
i) Which metal is likely to be Copper. Explain.
ii) Identify the strongest reducing agent.
iii) Which two half – cells would be combined to produce the highest voltage?.
iv) Give a cell representation of the cell in F (iii) above.
Date posted: May 22, 2019. Answers (1)
- Give any two uses of ethene gas(Solved)
Give any two uses of ethene gas
Date posted: May 22, 2019. Answers (1)
- The grid below represents part of the periodic table. Study it and answer the
questions that follow. (The letters are not the actual symbols of the...(Solved)
The grid below represents part of the periodic table. Study it and answer the
questions that follow. (The letters are not the actual symbols of the elements)

i) Select the element in period three which has the shortest atomic radius. Give a reason
for your answer.
ii) Using dots (.) and crosses (x) to represent outermost electrons, draw a diagram to
show the bonding in the compound formed when chlorine reacts with element X
iii) When three liters of chlorine gas were completely reacted with element Y, 11.85g of
the product were formed. Calculate the relative atomic mass of element Y
(R.A.M of chlorine = 35.5, molar gas volume = 24 liters)
Date posted: May 21, 2019. Answers (1)
- Use the information in the table below to answer the questions that follow
i) Are the members in this group likely to be conductor or non...(Solved)
Use the information in the table below to answer the questions that follow
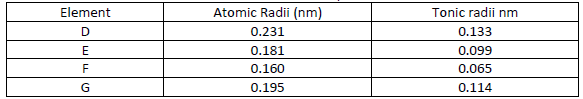
i) Are the members in this group likely to be conductor or non – conductors?
ii) Which element would have the lowest atomic number? Explain.
Date posted: May 21, 2019. Answers (1)
- A volume of 80cm3 of a mixture of propane (C3H8) and oxygen were ignited in an experiment.The products were cooled and passed through an aqueous...(Solved)
A volume of 80cm3 of a mixture of propane (C3H8) and oxygen were ignited in an experiment.The products were cooled and passed through an aqueous sodium hydroxide. The final volume
was reduced by 30cm3
a) Write the equation for the combustion of propane
b) Determine the volume of;
i) The component of the original mixture
ii) Residual oxygen
Date posted: May 21, 2019. Answers (1)
- Differentiate between empirical and molecular formula(Solved)
Differentiate between empirical and molecular formula
Date posted: May 21, 2019. Answers (1)