a) B: Because it requires little soap / less soap to form lather.
b) Sample C has temporary water hardness which is removed by boiling
Kavungya answered the question on May 23, 2019 at 09:19
-
Give two reasons why duralumin is preferred to aluminium in making aeroplane parts.
(Solved)
Give two reasons why duralumin is preferred to aluminium in making aeroplane parts.
Date posted:
May 23, 2019
.
Answers (1)
-
The table below gives three experiments on the reaction of excess sulphuric (VI) acid and 0.5g Zinc done under different conditions. In each case the...
(Solved)
The table below gives three experiments on the reaction of excess sulphuric (VI) acid and 0.5g Zinc done under different conditions. In each case the volume of gas liberated was recorded at different time intervals.
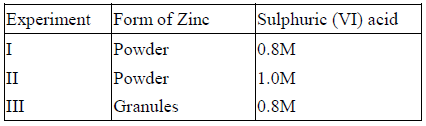
On the axes below, draw and label the three curves that would be obtained from the results above.

Date posted:
May 23, 2019
.
Answers (1)
-
Use the bond energies given below to calculate the heat of reaction for;
(Solved)
Use the bond energies given below to calculate the heat of reaction for;

Date posted:
May 23, 2019
.
Answers (1)
-
Study the diagram below then use it to answer the questions that follow.
a) Draw the wooden splint at the end of the experiment. If it...
(Solved)
Study the diagram below then use it to answer the questions that follow.

a) Draw the wooden splint at the end of the experiment. If it was slipped then removed.
b) Explain the appearance of the wooden splint in (a) above.
Date posted:
May 23, 2019
.
Answers (1)
-
Which is the chemical test to differentiate between alkenes and alkynes?
(Solved)
Which is the chemical test to differentiate between alkenes and alkynes?
Date posted:
May 22, 2019
.
Answers (1)
-
Use the table of logarithms to evaluate.
(Solved)
Use the table of logarithms to evaluate.

Date posted:
May 22, 2019
.
Answers (1)
-
The diagram below shows acidic and basic oxides fit in a general family of oxides.
a) State the name given to the type of oxides that...
(Solved)
The diagram below shows acidic and basic oxides fit in a general family of oxides.

a) State the name given to the type of oxides that would be placed in the shaded region.
b) Name two oxides that could be placed on the shaded region.
Date posted:
May 22, 2019
.
Answers (1)
-
Using well labeled diagrams, explain how water hardness can be removed by ion
exchange method.
(Solved)
Using well labeled diagrams, explain how water hardness can be removed by ion
exchange method.
Date posted:
May 22, 2019
.
Answers (1)
-
The solubility of copper (II) sulphate is 55g/100g of water at 75oC and 19g/100g of
water at 15oC. What mass of crystals would be deposited, if...
(Solved)
The solubility of copper (II) sulphate is 55g/100g of water at 75oC and 19g/100g of
water at 15oC. What mass of crystals would be deposited, if 150g of a saturated solution
is cooled from 75oC to 15oC.
Date posted:
May 22, 2019
.
Answers (1)
-
Reagent bottles labelled H2SO4 solution, K2CO3 solution and NaCl solution had labels
accidentally removed. A packet of blue litmus paper is lying near a long with...
(Solved)
Reagent bottles labelled H2SO4 solution, K2CO3 solution and NaCl solution had labels
accidentally removed. A packet of blue litmus paper is lying near a long with a rack of test-tubes, without using any other material explain how you would go about labeling the bottles correctly.
Date posted:
May 22, 2019
.
Answers (1)
-
The scheme below shows a reaction sequence starting with solid N. study it and answer
the questions that follows.
a) Name the cation present in solid N....
(Solved)
The scheme below shows a reaction sequence starting with solid N. study it and answer
the questions that follows.
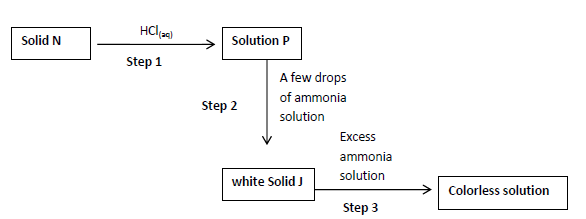
a) Name the cation present in solid N.
b) Write the formula of the complex ion in the colorless solution Q.
c) Write an ionic equation for the reaction in step 2.
Date posted:
May 22, 2019
.
Answers (1)
-
What is meant by the term strong acid?
(Solved)
What is meant by the term strong acid?
Date posted:
May 22, 2019
.
Answers (1)
-
The set-up below was used to prepare Nitric(V) acid in the laboratory.
a) Name liquid T.
b) Write an equation for the reaction taking place in the...
(Solved)
The set-up below was used to prepare Nitric(V) acid in the laboratory.
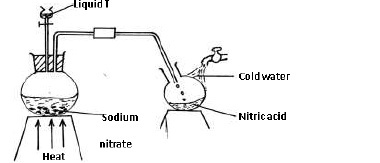
a) Name liquid T.
b) Write an equation for the reaction taking place in the flask.
c) State the reason why nitric (V) acid collected is brown in colour and explain how the
brown colour can be removed.
Date posted:
May 22, 2019
.
Answers (1)
-
The empirical formula of a compound is C2H5. When 11.6g of the compound was
allowed to evaporate; it occupied 4.8 dm3 at room temperature and pressure....
(Solved)
The empirical formula of a compound is C2H5. When 11.6g of the compound was
allowed to evaporate; it occupied 4.8 dm3 at room temperature and pressure. What is
its molecular formula? (MGV = 24dm3)
Date posted:
May 22, 2019
.
Answers (1)
-
A sample of unknown gas Z was shown by analysis to contain sulphur and oxygen.
The gas requires 28.3 seconds to diffuse through an aperture into...
(Solved)
A sample of unknown gas Z was shown by analysis to contain sulphur and oxygen.
The gas requires 28.3 seconds to diffuse through an aperture into a vacuum. An
identical amount of oxygen gas passes through the same aperture in 20 seconds.
Determine the molar mass of gas Z. (O=16, S=32).
Date posted:
May 22, 2019
.
Answers (1)
-
Write equations to show the effect of heat on each of the following.
(a) Sodium hydrogen carbonate.
(b) Silver nitrate
(c ) Sodium nitrate
(Solved)
Write equations to show the effect of heat on each of the following.
(a) Sodium hydrogen carbonate.
(b) Silver nitrate
(c ) Sodium nitrate
Date posted:
May 22, 2019
.
Answers (1)
-
When small amount of carbon (IV) oxide is passed through lime water, a white
precipitate is formed. When excess carbon (IV) oxide is bubbled through, the...
(Solved)
When small amount of carbon (IV) oxide is passed through lime water, a white
precipitate is formed. When excess carbon (IV) oxide is bubbled through, the white
precipitate dissolves to form a colorless solution.
a) Name the white precipitate.
b) Explain using chemical equations why the white precipitate dissolves in excess of
carbon(IV) oxide.
c) What will happen of the above colorless solution is boiled.
Date posted:
May 22, 2019
.
Answers (1)
-
When magnesium burns in air, it forms two products. When one of the products
dissolves in water, a colorless gas that turns red litmus paper blue...
(Solved)
When magnesium burns in air, it forms two products. When one of the products
dissolves in water, a colorless gas that turns red litmus paper blue is formed.
a) Name the product that dissolves in water to produce a colorless gas.
b) Write an equation for the formation of the colorless gas.
c) State any one use of the colorless gas.
Date posted:
May 22, 2019
.
Answers (1)
-
A form four student arranged the apparatus as shown below with the aim of collecting
dry hydrogen gas.
Identify with reasons two mistakes in the above set-up.
(Solved)
A form four student arranged the apparatus as shown below with the aim of collecting
dry hydrogen gas.
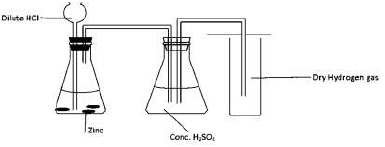
Identify with reasons two mistakes in the above set-up.
Date posted:
May 22, 2019
.
Answers (1)
-
The diagram below shows a Bunsen burner which is used as a source of heat in the
laboratory.
(Solved)
The diagram below shows a Bunsen burner which is used as a source of heat in the
laboratory.
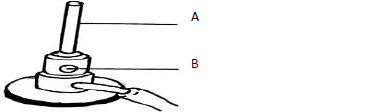
Name the parts labeled: (
A
B
Date posted:
May 22, 2019
.
Answers (1)