- Using reagents provided, explain how you could prepare dry zinc carbonate solid.
Zinc powder, dilute nitric (V) acid, Water and Solid sodium carbonate(Solved)
Using reagents provided, explain how you could prepare dry zinc carbonate solid.
Zinc powder, dilute nitric (V) acid, Water and Solid sodium carbonate
Date posted: August 14, 2019. Answers (1)
- In an experiment, iron was heated in the presence of excess oxygen and the following data was obtained.(Solved)
In an experiment, iron was heated in the presence of excess oxygen and the following data was obtained:
Mass of empty crucible = 10.98 g
Mass of crucible + iron before reaction = 12.66 g
Mass of crucible + iron after heating = 13.30 g
Calculate the empirical formula of the oxide of iron. (Fe =56, O = 16)
Date posted: August 14, 2019. Answers (1)
- Below is a list of oxides.(Solved)
Below is a list of oxides.

Select:
(a) A neutral oxide
(b) A highly water soluble basic oxide.
(c) An oxide which can react with sodium hydroxide and dilute hydrochloric acid.
Date posted: August 14, 2019. Answers (1)
- An oxide of Nitrogen was bubbled into a solution of sodium hydroxide in which phenolphthalein indicator has been added. The solution turned colourless after sometime.(Solved)
An oxide of Nitrogen was bubbled into a solution of sodium hydroxide in which phenolphthalein indicator has been added. The solution turned colourless after sometime.
(a) Name the oxide.
(b) Write an ionic equation for the reaction that took place.
Date posted: August 14, 2019. Answers (1)
- Study the flow chart below and answer the questions that follow.(Solved)
Study the flow chart below and answer the questions that follow.

(a) Identify N and P.
(b) Name the type of reaction in step 2.
Date posted: August 14, 2019. Answers (1)
- When heated in a current of nitrogen gas, magnesium reacts to form a compound.(Solved)
When heated in a current of nitrogen gas, magnesium reacts to form a compound.
(a) Write the chemical equation of the compound formed.
(b) Calculate the volume of nitrogen at s.t.p required to react with 0.72 g of magnesium.
(Mg = 24, molar gas volume at s.t.p = 22.4 dm3)
Date posted: August 14, 2019. Answers (1)
- An element P contains two isotopes
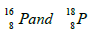 whose relative abundance is in the ratio 9:1. Find the relative atomic mass of P.(Solved)
whose relative abundance is in the ratio 9:1. Find the relative atomic mass of P.(Solved)
An element P contains two isotopes 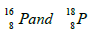 whose relative abundance is in the ratio 9:1. Find the relative atomic mass of P.
whose relative abundance is in the ratio 9:1. Find the relative atomic mass of P.
Date posted: August 14, 2019. Answers (1)
- Study the equation below and answer the question that follows.(Solved)
Study the equation below and answer the question that follows.
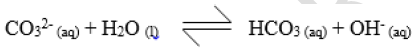
Identify the acid in the forward reaction. Give a reason for your answer.
Date posted: August 14, 2019. Answers (1)
- A 10 cm3 volume concentrated hydrochloric acid of density 1.18 g/cm3 and purity of 36 percent dissolved in 250 cm3 of distilled water. Calculate the...(Solved)
A 10 cm3 volume concentrated hydrochloric acid of density 1.18 g/cm3 and purity of 36 percent dissolved in 250 cm3 of distilled water. Calculate the morality of the solution. (H = 1, Cl = 35.5)
Date posted: August 14, 2019. Answers (1)
- Archeologists can determine the age of organic matter by measuring the proportion of carbon-14 present in a sample. Assuming that carbon-14 has a half- life...(Solved)
Archeologists can determine the age of organic matter by measuring the proportion of carbon-14 present in a sample. Assuming that carbon-14 has a half- life of 5600 years, calculate the age of a piece of wood found to contain 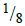 as much carbon-14 as in a living material.
as much carbon-14 as in a living material.
Date posted: August 14, 2019. Answers (1)
- Explain the observation made in the experiment whose set up is shown below.(Solved)
Explain the observation made in the experiment whose set up is shown below.
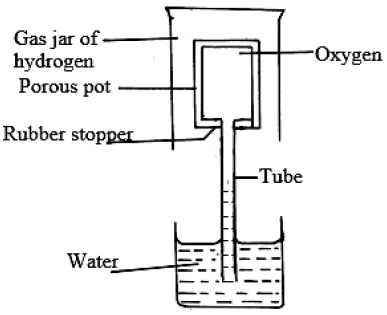
Date posted: August 14, 2019. Answers (1)
- Element G and T have atomic number 20 and 9, respectively.(Solved)
Element G and T have atomic number 20 and 9, respectively.
(a) Write down the electron arrangement of the ion of G.
(b) Which element has the same electron arrangement as the ion of T?
(c) Write the formula of compound formed by reaction of G and T.
Date posted: August 14, 2019. Answers (1)
- An Element X has first, second and third ionization energies. State and explain how their trends compare.(Solved)
An Element X has first, second and third ionization energies. State and explain how their trends compare.
Date posted: August 5, 2019. Answers (1)
- Why is an atom said to be electrically neutral?(Solved)
Why is an atom said to be electrically neutral?
Date posted: August 5, 2019. Answers (1)
- Name one ore of Zinc metal(Solved)
Name one ore of Zinc metal
Date posted: August 5, 2019. Answers (1)
- A sample of a colorless solution is suspected to be Zinc (II) sulphate. Describe some tests that can be carried to prove this.(Solved)
A sample of a colorless solution is suspected to be Zinc (II) sulphate. Describe some tests that can be carried to prove this.
Date posted: August 5, 2019. Answers (1)
- Calculate the percentage of nitrogen in calcium nitrate(Solved)
Calculate the percentage of nitrogen in calcium nitrate
Date posted: August 5, 2019. Answers (1)
- Dry carbon (II) oxide gas reacts with hot lead (II) oxide as shown in the equation below.(Solved)
Dry carbon (II) oxide gas reacts with hot lead (II) oxide as shown in the equation below.
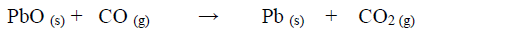
a) Name another gas that can be used to function as carbon (II) oxide in this experiment.
b) With an appropriate reason, identify the oxidizing agent in the equation above.
Date posted: August 5, 2019. Answers (1)
- Highlight one precaution observed in each of the following cases:
i. When evaporating Ethanol.
ii. When heating to dryness a hydrated salts.(Solved)
Highlight one precaution observed in each of the following cases:
i. When evaporating Ethanol.
ii. When heating to dryness a hydrated salts.
Date posted: August 5, 2019. Answers (1)
- A metal Y with atomic number 11 burns in chlorine to produce a white solid X.Describe the following properties of X in terms of
i)...(Solved)
A metal Y with atomic number 11 burns in chlorine to produce a white solid X.Describe the following properties of X in terms of
i) Solubility.
ii).Electrical conductivity
Date posted: August 5, 2019. Answers (1)