(i) 100 – 1100C
(ii) Impure - it melts and boils over a range of temperature.
(iii) - Increases the boiling point.
maurice.mutuku answered the question on September 12, 2019 at 05:12
-
The curves below represent the volume of carbon (IV) oxide gas evolved when 1M concentrated hydrochloric acid and 60g of calcium carbonate and also 0.5M...
(Solved)
The curves below represent the volume of carbon (IV) oxide gas evolved when 1M concentrated hydrochloric acid and 60g of calcium carbonate and also 0.5M concentrated hydrochloric acid was reacted with the same quantity of the carbonate.

(a) Which of the curves represent the reaction of 1M concentrated hydrochloric acid with powdered calcium carbonate.
Explain.
(b) Explain why the curves flatters at the same level of production of carbon (IV) oxide.
Date posted:
September 12, 2019
.
Answers (1)
-
Xg of sodium hydroxide were dissolved in distilled water to make 100cm3 of solution 40cm3 of this solution required 25cm3 of 1M sulphuric (VI) acid....
(Solved)
Xg of sodium hydroxide were dissolved in distilled water to make 100cm3 of solution 40cm3 of this solution required 25cm3 of 1M sulphuric (VI) acid. Determine the mass of X of solution of sodium hydroxide (Na = 23, O = 16, H = 1)
Date posted:
September 11, 2019
.
Answers (1)
-
Calculate the enthalpy of formation of propane.
(Solved)
Given that:
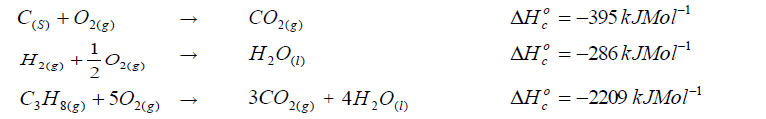
Calculate the enthalpy of formation of propane.
Date posted:
September 11, 2019
.
Answers (1)
-
The following set-up was used to prepare Carbon (II) Oxide in laboratory.
(Solved)
The following set-up was used to prepare Carbon (II) Oxide in laboratory.

(a) Name liquid X and state its value.
(b) Explain why the gas is collected over water.
Date posted:
September 11, 2019
.
Answers (1)
-
Using relevant equations show that zinc oxide is an amphoteric oxide.
(Solved)
Using relevant equations show that zinc oxide is an amphoteric oxide.
Date posted:
September 11, 2019
.
Answers (1)
-
The diagram below represents a flame of the Bunsen burner.
(Solved)
The diagram below represents a flame of the Bunsen burner.

A piece of paper is flipped over the flame as shown in the diagram. Draw a sketch to show the outcome.
Date posted:
September 11, 2019
.
Answers (1)
-
The chart below represents the extraction of iron and some of its uses.
(Solved)
The chart below represents the extraction of iron and some of its uses.


(a) Name the raw materials fed into the blast furnace.
(b) Name 3 exhaust gases emitted from the blast furnace.
(c) (i) Why is it necessary to convert pig iron into wrought iron
(ii) State one commercial use of iron.
(d) Name substances A,B,C,X,Y
(e) i) Write equations for reactions in steps II and II
ii) Write an ionic equation for the reaction in step I.
iii) What observations are made in steps I and II?
Date posted:
September 11, 2019
.
Answers (1)
-
The enthalpies of combustion of carbon, hydrogen and ethanol are given below.
(Solved)
The enthalpies of combustion of carbon, hydrogen and ethanol are given below.
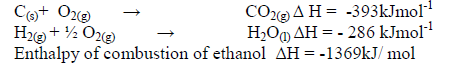
i) Draw an energy cycle diagram that links the enthalpy of formation of ethanol to enthalpies of combustion of Carbon,hydrogen and ethanol.
ii) Determine the enthalpy of formation of ethanol
Date posted:
September 11, 2019
.
Answers (1)
-
A fixed mass of gas occupies 105cm3 at -140C and 650mmHg pressure. At what temperature will it have a volume of 15cm3 if the pressure...
(Solved)
A fixed mass of gas occupies 105cm3 at -140C and 650mmHg pressure. At what temperature will it have a volume of 15cm3 if the pressure is adjusted to 690 mmHg pressure
Date posted:
September 11, 2019
.
Answers (1)
-
Give a reason why the formula mass of NO2 is sometimes 92 instead of 46.
(Solved)
Give a reason why the formula mass of NO2 is sometimes 92 instead of 46.
Date posted:
September 10, 2019
.
Answers (1)
-
Consider the chromatogram below.
(Solved)
Consider the chromatogram below.

A piece of chromatogram paper was spotted with colour inks obtained from pens labeled A to F. The diagram above shows the spots after the chromatograph was developed.
(a) Which two pens contained the same pigment?
(b) According to the chromatogram which pigments are present in the inks of the pen number F
(c) Describe how one could get a sample of yellow pigment
Date posted:
September 10, 2019
.
Answers (1)
-
Hydrazine gas, shown below, burns in oxygen to form nitrogen gas and steam.
(Solved)
Hydrazine gas, shown below, burns in oxygen to form nitrogen gas and steam.

(a) Write an equation for the reaction
(b) Using the bond energies given below, calculate the enthalpy change for the reaction in (a) above

Date posted:
September 10, 2019
.
Answers (1)
-
A form one class carried out an experiment to determine the active part of air. The diagram below shows the set-up of the experiment and...
(Solved)
A form one class carried out an experiment to determine the active part of air. The diagram below shows the set-up of the experiment and also the observation made.

(a) Identify substance M
(b) State two reasons for the suitability of substance M for this experiment
(c) Write the equation for the reaction of substance M and the active part of air
Date posted:
September 10, 2019
.
Answers (1)
-
Draw a dot and cross diagram to show bonding in sulphur (IV) oxide
(Solved)
Draw a dot and cross diagram to show bonding in sulphur (IV) oxide
Date posted:
September 10, 2019
.
Answers (1)
-
Give one use of charcoal in the sugar refinery industry.
(Solved)
Give one use of charcoal in the sugar refinery industry.
Date posted:
September 10, 2019
.
Answers (1)
-
Study the flow chart below and answer the questions that follow.
(Solved)
Study the flow chart below and answer the questions that follow.

(a) Identify solid G
(b) Write a balanced chemical equation between the yellow solid and dilute nitric acid.
(c) Write the formula of the complex ion in solution F
Date posted:
September 10, 2019
.
Answers (1)
-
The diagram below shows a set-up of apparatus that can be used to prepare nitrogen (IV) oxide. Study it and use it to answer the...
(Solved)
The diagram below shows a set-up of apparatus that can be used to prepare nitrogen (IV) oxide. Study it and use it to answer the questions that follow.
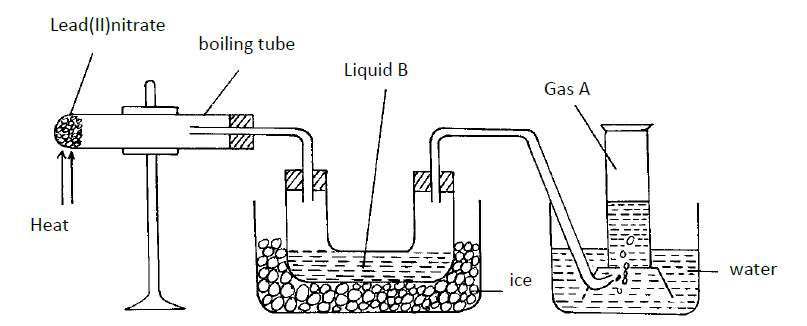
(i) Write the equation for the reaction that takes place in the boiling tube.
(ii) State the observations made in the boiling tube.
(iii) Explain why lead (II) nitrate is preferred over other metal nitrates in this experiment.
(iv) Describe how gas A can be identified.
(b) (i) Name liquid B
(ii) Write a chemical equation to show how liquid B is formed in this experiment.
(c) (i) In another experiment, excess aqueous lead (II) nitrate solution was reacted with a solution which contained 2.34g of sodium chloride. Calculate the mass of precipitate formed in this reaction. (Pb = 207, Cl = 35.5, Na = 23)
(ii) Write an ionic equation for the reaction that takes place when nitrogen (IV) oxide reacts with aqueous sodium hydroxide.
Date posted:
September 10, 2019
.
Answers (1)
-
The flow chart below shows some processes in the extraction of zinc. Study it and answer the questions that follow.
(Solved)
The flow chart below shows some processes in the extraction of zinc. Study it and answer the questions that follow.
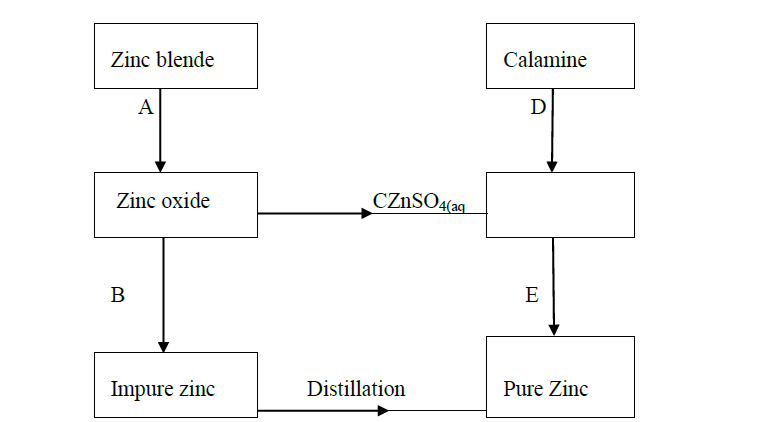
(a) Name the processes represented by A and E.
(b) State the reagents required for processes B, C and D.
(c) Write a chemical equation of the reaction that occurs in process B.
(d) With an aid of a diagram, explain how you would obtain a pure sample of zinc by process E
(e) State two commercial uses of zinc metal.
Date posted:
September 10, 2019
.
Answers (1)
-
The enthalpies of combustion of calcium, carbon and decomposition of calcium carbonate are indicated below;
(Solved)
The enthalpies of combustion of calcium, carbon and decomposition of calcium carbonate are indicated below;
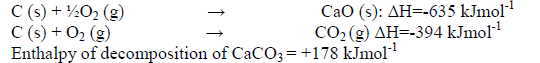
(i) Draw an energy cycle diagram that links the enthalpy of formation of calcium carbonate to enthalpies of combustion of calcium, carbon and decomposition of calcium carbonate.
(ii) Determine the enthalpy of formation of calcium carbonate.
Date posted:
September 10, 2019
.
Answers (1)
-
An aqueous solution of zinc sulphate is electrolysed using platinum electrodes as shown in the set up below.
(Solved)
An aqueous solution of zinc sulphate is electrolysed using platinum electrodes as shown in the set up below.
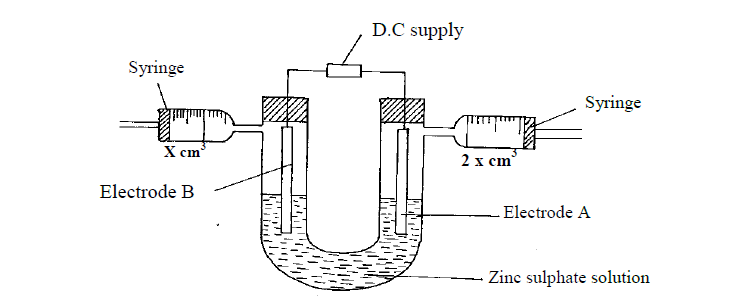
(a) (i) Write a half equation for the reaction taking place at electrode A.
(ii) Identify electrodes A and B
(iii) State and explain the observation at electrode B if copper plate was used instead of platinum electrode.
Date posted:
September 10, 2019
.
Answers (1)