(i) NH4NO3(s)-------->N2O(g) + 2H2O(g)
(ii) NH4NO3 should not be heated further if the quantity remaining is small because it may explode or A mixture of NH4Cl & KNO3 can be used instead of NH4NO3 leading to double decomposition taking place safely without explosion
(iii) An hydrous calcium chloride in a u-tube
(iv) Reacts with oxygen to form brown fumes of Nitrogen (IV) Oxide
2N2O(g) + O2(g)------>2NO2(g)
(v) – Has no colour
- Has a slight sweet smell
(vi) Fairly soluble in water
- Denser than air
maurice.mutuku answered the question on October 22, 2019 at 09:25
- The chart below shows a summary for the preparation of nitrogen gas from air(Solved)
The chart below shows a summary for the preparation of nitrogen gas from air

(a) What is the purpose of the sodium hydroxide?
(b) Write an equation for the reaction taking place in chamber II
(c) The nitrogen gas obtained is not pure. Explain
Date posted: October 22, 2019. Answers (1)
- A mixture of N2, H2 and NH3 was bubbled through 0.2M hydrochloric acid solution.
The final concentration of the acid was found to be 0.1M. Give...(Solved)
A mixture of N2, H2 and NH3 was bubbled through 0.2M hydrochloric acid solution.
The final concentration of the acid was found to be 0.1M. Give explanation
Date posted: October 22, 2019. Answers (1)
- Study the flow charts below and use them to answer the questions that follow:(Solved)
Study the flow charts below and use them to answer the questions that follow:
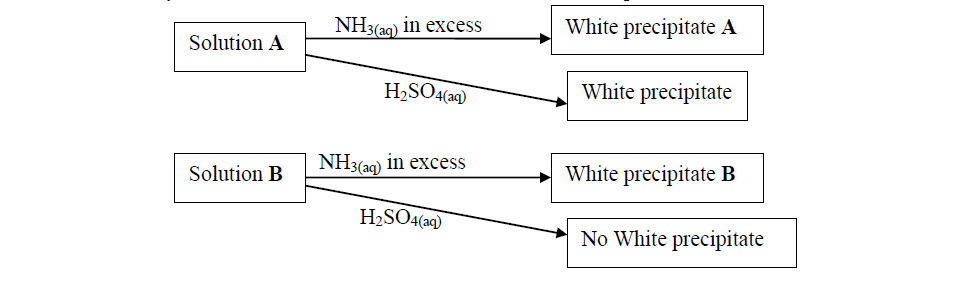
(a) Identify possible cations present in:
(i) Solution A
(ii) Solution B
(b) State and explain the observations made when a sample of dry white precipitate B is heated in a test-tube
Date posted: October 22, 2019. Answers (1)
- The diagram below is used in preparation of a gas in the laboratory. Answer the questions that follow;(Solved)
The diagram below is used in preparation of a gas in the laboratory. Answer the questions that follow;

(a) Name gas X
(b) State one physical property which makes it possible for the gas to be collected as shown
(c) State one commercial use of gas X
Date posted: October 22, 2019. Answers (1)
- (a) Explain the importance of the high percentage of nitrogen in air(b) Why is nitrogen used for storage of semen in artificial insemination?(Solved)
(a) Explain the importance of the high percentage of nitrogen in air
(b) Why is nitrogen used for storage of semen in artificial insemination?
Date posted: October 22, 2019. Answers (1)
- Ammonia gas is prepared in the laboratory by the action of an alkali on an ammonium salt.
A student wanted to prepare a sample of ammonia...(Solved)
Ammonia gas is prepared in the laboratory by the action of an alkali on an ammonium salt.
A student wanted to prepare a sample of ammonia gas in the laboratory.
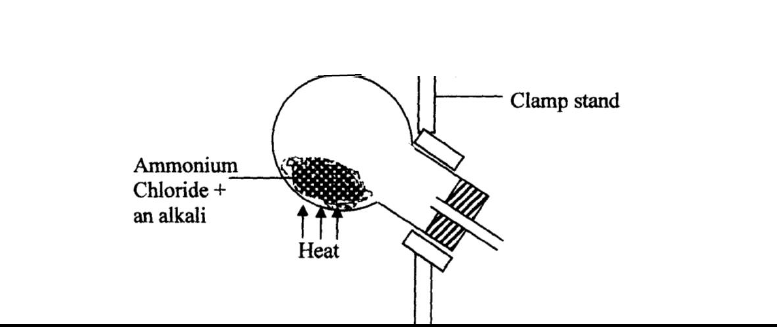
(a) Give one alkali that can be used in the above experiment
(b) Write an equation for the reaction that takes place in the above experiment
Date posted: October 22, 2019. Answers (1)
- The diagram below shows the catalytic oxidation of ammonia gas. Use it to answer the questions that follow:-(Solved)
The diagram below shows the catalytic oxidation of ammonia gas. Use it to answer the questions that follow:-
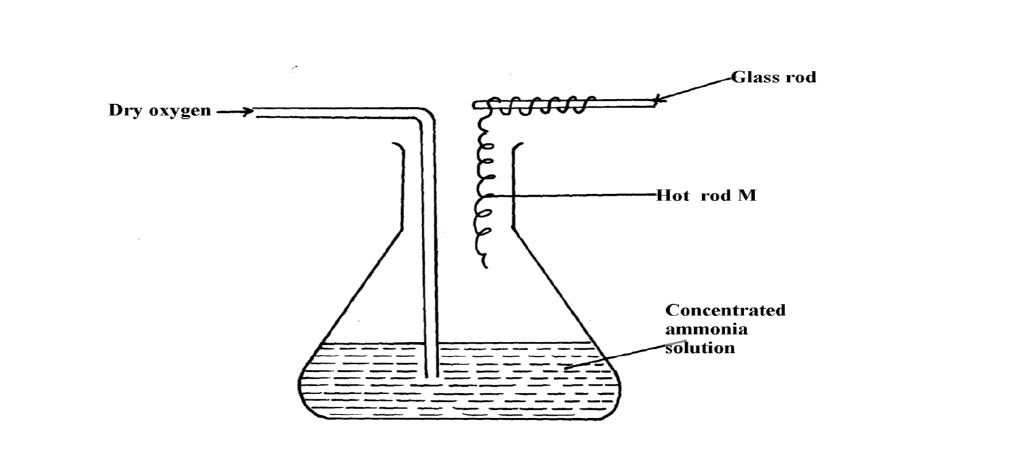
(a) What metal could rod M be made of?
(b) State and explain two observations made inside the conical flask
Date posted: October 22, 2019. Answers (1)
- A hydrocarbon compound Z decolourizes bromine liquid in the presence of light but does not decolourize acidified potassium manganate (VII). Name and draw the structural...(Solved)
A hydrocarbon compound Z decolourizes bromine liquid in the presence of light but does not decolourize acidified potassium manganate (VII). Name and draw the structural formula of the eighth member of this homologous series
Date posted: October 22, 2019. Answers (1)
- Write down all the isomers of but-z-ene and give their IUPAC names(Solved)
Write down all the isomers of but-z-ene and give their IUPAC names
Date posted: October 22, 2019. Answers (1)
- (i) 2.63g of a solution of sodium chloride at 20.0oC was reacted with silver nitrate. After filtration,washing and drying, 2.36g of silver chloride was obtained....(Solved)
(i) 2.63g of a solution of sodium chloride at 20.0oC was reacted with silver nitrate. After filtration,washing and drying, 2.36g of silver chloride was obtained. Determine the solubility of sodium
chloride at 20.0oC . (Na=23, Cl= 35.5, Ag = 108)
(ii) Determine the number of moles of carbon (IV) Oxide gas produced when sodium carbonate reacted with dilute sulphuric (VI) acid (Molar gas volume =24dm3)
Date posted: October 22, 2019. Answers (1)
- Substance “M” with a general formula C2Hy burnt in chlorine gas with a red flame producing a cloud of black specks and colourless gas G.
(a)...(Solved)
Substance “M” with a general formula C2Hy burnt in chlorine gas with a red flame producing a cloud of black specks and colourless gas G.
(a) State the collective name for compounds which ‘M’ belongs
(b) With reason, state the identity of the black specks and colour gas “G”.
Date posted: October 22, 2019. Answers (1)
- Study the data given in the following table and answer the questions that follow. The letters are not the actual symbols of elements.(Solved)
Study the data given in the following table and answer the questions that follow. The letters are not the actual symbols of elements.
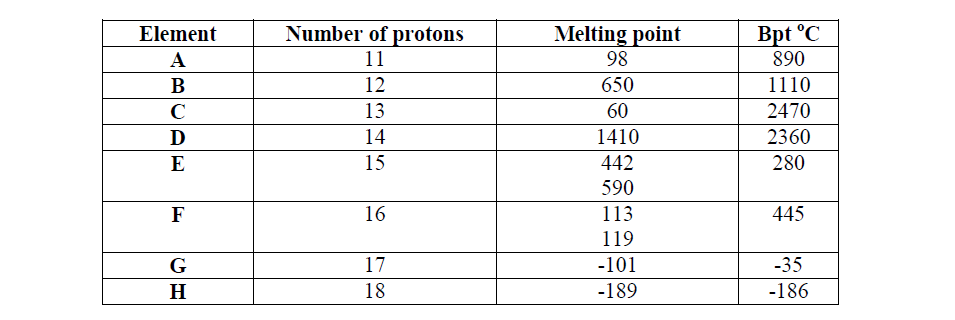
(i) State and explain the trend in melting point in A B C
(ii) Explain why the melting point and boiling points of element D is the highest
(iii) Explain why the element represented by letter E has two melting point values
(iv) Write down the chemical formula between element C and sulphate ions
(v) Name the chemical family in which H belong and state one use of the element
(vi) What is the nature of the oxide of the elements represented by letters C and F?
Date posted: October 22, 2019. Answers (1)
- Give the systematic names of the following compounds;(Solved)
Give the systematic names of the following compounds;

Date posted: October 22, 2019. Answers (1)
- Study the scheme below and answer the questions that follow:(Solved)
Study the scheme below and answer the questions that follow:

Name the reagents in
Step I
Step II
Step IV
(ii) Write an equation for the complete combustion of 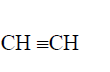
(iii) Give two uses of CH4
Date posted: October 22, 2019. Answers (1)
- Ethene is used in making polyethene bag in a process called polymerization
(i) Name the type of polymer that is formed when ethane polymerise
(ii) Describe a...(Solved)
Ethene is used in making polyethene bag in a process called polymerization
(i) Name the type of polymer that is formed when ethane polymerise
(ii) Describe a simple chemical test that can be used to identify ethane gas in the laboratory
Date posted: October 22, 2019. Answers (1)
- Give the names of the following:
(i) CH3CH2CH3
(ii) CH3CCCH3(Solved)
Give the names of the following:
(i) CH3CH2CH3
(ii) CH3CCCH3
Date posted: October 22, 2019. Answers (1)
- Name the following compounds:(CH3)3 C CH2 CH2 CH3(Solved)
Name the following compounds:(CH3)3 C CH2 CH2 CH3
Date posted: October 22, 2019. Answers (1)
- Give two ways how the disposal of polymers such as polychloroethene by burning pollutes the environment(Solved)
Give two ways how the disposal of polymers such as polychloroethene by burning pollutes the environment
Date posted: October 22, 2019. Answers (1)
- State the observations made when buton-l-ol reacts with:-
(i) Acidified potassium dichromate (VI) solution
(ii) Potassium metal(Solved)
State the observations made when buton-l-ol reacts with:-
(i) Acidified potassium dichromate (VI) solution
(ii) Potassium metal
Date posted: October 22, 2019. Answers (1)
- Give the systematic names of the following compounds:-(Solved)
Give the systematic names of the following compounds:-

Date posted: October 22, 2019. Answers (1)