- CuO is reduced to Copper metal
- NH3 is oxidized to water and nitrogen
maurice.mutuku answered the question on October 24, 2019 at 05:40
- State the conditions under which nitrogen react with hydrogen to form ammonia during Haber process(Solved)
State the conditions under which nitrogen react with hydrogen to form ammonia during Haber process
Date posted: October 24, 2019. Answers (1)
- When a few drops of aqueous ammonia were added to a colourless solution X, a white precipitate was formed. On addition of more aqueous ammonia,...(Solved)
When a few drops of aqueous ammonia were added to a colourless solution X, a white precipitate was formed. On addition of more aqueous ammonia, the white precipitate dissolved to a colourless solution Q
(a) Name the white precipitate formed
(b) Write formula of the complex ion present in the colourless solution Q
(c) Write an ionic equation for the formation of the white precipitate
Date posted: October 24, 2019. Answers (1)
- Ammonia turns wet red litmus paper blue. Which ion is responsible for this reaction(Solved)
Ammonia turns wet red litmus paper blue. Which ion is responsible for this reaction
Date posted: October 24, 2019. Answers (1)
- Nitrogen (I) Oxide supports, combustion of burning charcoal. Write an equation to show this reaction(Solved)
Nitrogen (I) Oxide supports, combustion of burning charcoal. Write an equation to show this reaction
Date posted: October 24, 2019. Answers (1)
- When sulphur powder is heated to over 400oC the following changes are observed:-
At 113oC it melts into light brown liquid. The liquid then darkens to...(Solved)
When sulphur powder is heated to over 400oC the following changes are observed:-
At 113oC it melts into light brown liquid. The liquid then darkens to become reddish-brown
and very viscous at 160oC. Above 160oC the liquid becomes almost black. At the boiling point the liquid becomes mobile. Explain these observations
Date posted: October 24, 2019. Answers (1)
- The diagram below was used to investigate the reaction between nitrogen(I)oxide and copper turnings. Study it and answer the questions that follow:(Solved)
The diagram below was used to investigate the reaction between nitrogen(I)oxide and copper turnings. Study it and answer the questions that follow:

a) What has been omitted in the set-up?
b) Write a chemical equation for the reaction that took place in the combustion tube
c) State one use of gas P
Date posted: October 24, 2019. Answers (1)
- The diagram below represents an in complete set-up for preparation of a dry sample of gas R(Solved)
The diagram below represents an in complete set-up for preparation of a dry sample of gas R

a) Complete the set-up to show how a dry sample of gas R is collected
b) Write a chemical equation for the reaction that produces gas R
Date posted: October 22, 2019. Answers (1)
- The diagram below is a set-up used in preparation of ammonia solution. Study it and answer the questions that follow(Solved)
The diagram below is a set-up used in preparation of ammonia solution. Study it and answer the questions that follow

(i) What is the purpose of the filter funnel in the set-up above?
(ii) What would happen if a delivery tube was used in place of the filter funnel?
(iii) What observation would be made on litmus paper placed into the solution in the beaker at the end of the experiment?
Date posted: October 22, 2019. Answers (1)
- The diagram below is a set-up for preparation and collection of a gas. Study it answer the questions that follow:(Solved)
The diagram below is a set-up for preparation and collection of a gas. Study it answer the questions that follow:

(i) Identify gas X
(ii) Write an equation for the formation of gas X
(iii) What precaution should be observed when preparing gas X by the above method?
(iv) Describe the suitable drying agent for gas X
(v) How can one confirm that the gas collected is gas X?
(vi) State two physical properties of gas X
Date posted: October 22, 2019. Answers (1)
- The chart below shows a summary for the preparation of nitrogen gas from air(Solved)
The chart below shows a summary for the preparation of nitrogen gas from air

(a) What is the purpose of the sodium hydroxide?
(b) Write an equation for the reaction taking place in chamber II
(c) The nitrogen gas obtained is not pure. Explain
Date posted: October 22, 2019. Answers (1)
- A mixture of N2, H2 and NH3 was bubbled through 0.2M hydrochloric acid solution.
The final concentration of the acid was found to be 0.1M. Give...(Solved)
A mixture of N2, H2 and NH3 was bubbled through 0.2M hydrochloric acid solution.
The final concentration of the acid was found to be 0.1M. Give explanation
Date posted: October 22, 2019. Answers (1)
- Study the flow charts below and use them to answer the questions that follow:(Solved)
Study the flow charts below and use them to answer the questions that follow:
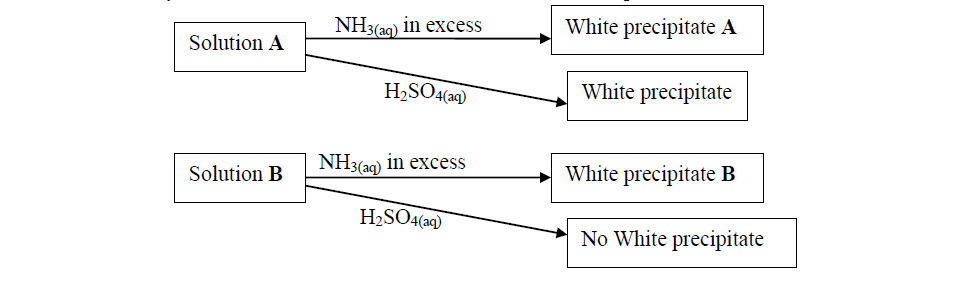
(a) Identify possible cations present in:
(i) Solution A
(ii) Solution B
(b) State and explain the observations made when a sample of dry white precipitate B is heated in a test-tube
Date posted: October 22, 2019. Answers (1)
- The diagram below is used in preparation of a gas in the laboratory. Answer the questions that follow;(Solved)
The diagram below is used in preparation of a gas in the laboratory. Answer the questions that follow;

(a) Name gas X
(b) State one physical property which makes it possible for the gas to be collected as shown
(c) State one commercial use of gas X
Date posted: October 22, 2019. Answers (1)
- (a) Explain the importance of the high percentage of nitrogen in air(b) Why is nitrogen used for storage of semen in artificial insemination?(Solved)
(a) Explain the importance of the high percentage of nitrogen in air
(b) Why is nitrogen used for storage of semen in artificial insemination?
Date posted: October 22, 2019. Answers (1)
- Ammonia gas is prepared in the laboratory by the action of an alkali on an ammonium salt.
A student wanted to prepare a sample of ammonia...(Solved)
Ammonia gas is prepared in the laboratory by the action of an alkali on an ammonium salt.
A student wanted to prepare a sample of ammonia gas in the laboratory.
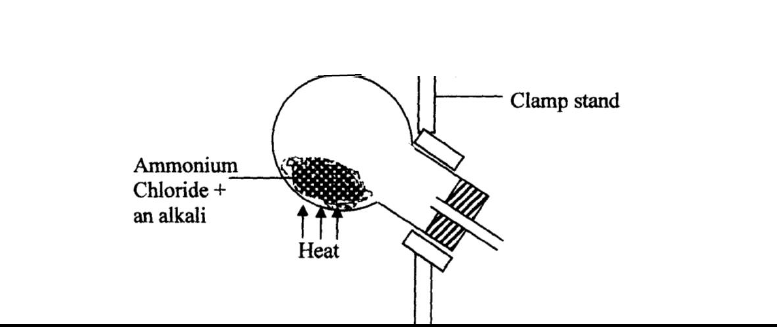
(a) Give one alkali that can be used in the above experiment
(b) Write an equation for the reaction that takes place in the above experiment
Date posted: October 22, 2019. Answers (1)
- The diagram below shows the catalytic oxidation of ammonia gas. Use it to answer the questions that follow:-(Solved)
The diagram below shows the catalytic oxidation of ammonia gas. Use it to answer the questions that follow:-
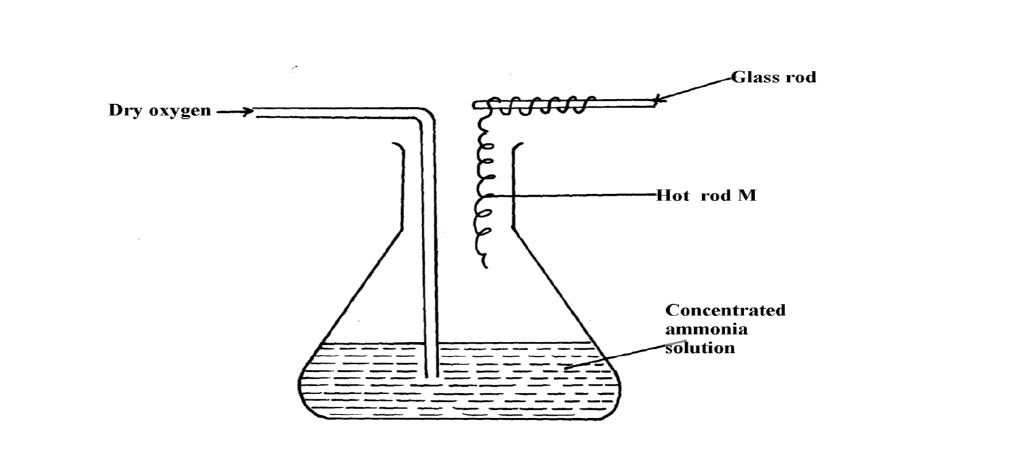
(a) What metal could rod M be made of?
(b) State and explain two observations made inside the conical flask
Date posted: October 22, 2019. Answers (1)
- A hydrocarbon compound Z decolourizes bromine liquid in the presence of light but does not decolourize acidified potassium manganate (VII). Name and draw the structural...(Solved)
A hydrocarbon compound Z decolourizes bromine liquid in the presence of light but does not decolourize acidified potassium manganate (VII). Name and draw the structural formula of the eighth member of this homologous series
Date posted: October 22, 2019. Answers (1)
- Write down all the isomers of but-z-ene and give their IUPAC names(Solved)
Write down all the isomers of but-z-ene and give their IUPAC names
Date posted: October 22, 2019. Answers (1)
- (i) 2.63g of a solution of sodium chloride at 20.0oC was reacted with silver nitrate. After filtration,washing and drying, 2.36g of silver chloride was obtained....(Solved)
(i) 2.63g of a solution of sodium chloride at 20.0oC was reacted with silver nitrate. After filtration,washing and drying, 2.36g of silver chloride was obtained. Determine the solubility of sodium
chloride at 20.0oC . (Na=23, Cl= 35.5, Ag = 108)
(ii) Determine the number of moles of carbon (IV) Oxide gas produced when sodium carbonate reacted with dilute sulphuric (VI) acid (Molar gas volume =24dm3)
Date posted: October 22, 2019. Answers (1)
- Substance “M” with a general formula C2Hy burnt in chlorine gas with a red flame producing a cloud of black specks and colourless gas G.
(a)...(Solved)
Substance “M” with a general formula C2Hy burnt in chlorine gas with a red flame producing a cloud of black specks and colourless gas G.
(a) State the collective name for compounds which ‘M’ belongs
(b) With reason, state the identity of the black specks and colour gas “G”.
Date posted: October 22, 2019. Answers (1)