 Get premium membership
Get premium membership and access questions with answers, video lessons as well as revision papers.
- Describe how a solid mixture of Zinc sulphate and lead (ii) Sulphate can be separated into Solid samples.(Solved)
Describe how a solid mixture of Zinc sulphate and lead (ii) Sulphate can be separated into Solid samples.
Date posted: September 17, 2019.
- A globe representing the earth has a radius of 0.5m. point A(00, 100W), B (00, 350E), P(600N, 1100E) and Q(600N, 1200W) are marked on...(Solved)
A globe representing the earth has a radius of 0.5m. point A(00, 100W), B (00, 350E), P(600N, 1100E) and Q(600N, 1200W) are marked on the globe.
a)Find the length of arc AB, leaving your answer in term of π
b)If O is the centre of the latitude 600N, find the area of the minor sector OPQ
c)Of the local time at Q is 10.30 a.m. on Monday, Determine the local time and day at P.
Date posted: September 17, 2019.
- Hydrogen Chloride gas is a colourless gas which dissolves readily in water forming hydrochloric acid.
(Solved)
(i) At room temperature and pressure, 1.00dm3 of water dissolves 432 dm3 of hydrogen chloride gas. How many moles of hydrogen chloride dissolves in 1dm3 water (1 mole at r.t.p. occupies 24.0dm3)
(ii) The hydrochloric acid formed has a volume of 1.40 dm3; calculate the concentration of the acid in mol/dm3.
Date posted: September 17, 2019.
- The pH values of some solutions labeled K,H,L,P and R are given in the table below. Use the information to answer the questions that follow.(Solved)
The pH values of some solutions labeled K,H,L,P and R are given in the table below. Use the information to answer the questions that follow.
 (a)Identify the solution with the highest concentration of hydroxyl ions.
(b)Which solution is likely to be sodium chloride solution?
(c)Which solution would react most vigorously with magnesium metal?
(a)Identify the solution with the highest concentration of hydroxyl ions.
(b)Which solution is likely to be sodium chloride solution?
(c)Which solution would react most vigorously with magnesium metal?
Date posted: September 17, 2019.
- The table below shows the current taxation rates in Kenya
A member of parliament’s monthly earnings are as follows: Basic salary Kshs. 300 000, commuted mileage...(Solved)
The table below shows the current taxation rates in Kenya
 A member of parliament’s monthly earnings are as follows: Basic salary Kshs. 300 000, commuted mileage allowance of Kshs. 75 000, entertain allowance Ksh. 60 000, extraneous allowance Ksh. 30 000, house allowance Kshs. 70,000, car maintaince allowance Kshs 247 000. he is entitled to a personal relief of Kshs. 1162 every month.
a)If before the promulgation of the new constitution all the MP’s allowances were tax free, calculate the PAYE the MP’s used to pay
b)In the new constitution all the MP’s are taxed just like the other Kenyans. Calculate the percentage increase in the MP’s PAYE after the new law was passed.
A member of parliament’s monthly earnings are as follows: Basic salary Kshs. 300 000, commuted mileage allowance of Kshs. 75 000, entertain allowance Ksh. 60 000, extraneous allowance Ksh. 30 000, house allowance Kshs. 70,000, car maintaince allowance Kshs 247 000. he is entitled to a personal relief of Kshs. 1162 every month.
a)If before the promulgation of the new constitution all the MP’s allowances were tax free, calculate the PAYE the MP’s used to pay
b)In the new constitution all the MP’s are taxed just like the other Kenyans. Calculate the percentage increase in the MP’s PAYE after the new law was passed.
Date posted: September 17, 2019.
- Use the diagram below to answer the questions that follow:(Solved)
Use the diagram below to answer the questions that follow:
 a) Identify the radiations
A
C
b)The table below gives the rate of decay for a sample of radioactive element K. Study it and answer the question that follows:
a) Identify the radiations
A
C
b)The table below gives the rate of decay for a sample of radioactive element K. Study it and answer the question that follows:
 Determine the half-life of element K.
Determine the half-life of element K.
Date posted: September 17, 2019.
- The diagram below represents the urinary system of a mammal. Study it carefully and answer the questions that follow.(Solved)
The diagram below represents the urinary system of a mammal. Study it carefully and answer the questions that follow.
 a) Name the parts labeled C, D and E.
b) i) Identify the part labeled A.
ii) State the hormone produced by the part you have identified in b(i) above and its function.
c) Which of the blood vessels (B and F) contains:
i) A higher concentration of urea.
ii) Lower concentration of oxygen.
d) State how the nephrons of desert mammals are structurally adapted.
a) Name the parts labeled C, D and E.
b) i) Identify the part labeled A.
ii) State the hormone produced by the part you have identified in b(i) above and its function.
c) Which of the blood vessels (B and F) contains:
i) A higher concentration of urea.
ii) Lower concentration of oxygen.
d) State how the nephrons of desert mammals are structurally adapted.
Date posted: September 17, 2019.
- The line PQ is 6cm long. On one side of the line,
a)Draw the locus of T such that the area of triangle PTQ =...(Solved)
The line PQ is 6cm long. On one side of the line,
a)Draw the locus of T such that the area of triangle PTQ = 12cm2
b)Determine two points on the locus obtained in part a) above such that angle PTQ = 700 and
label them as T1 and T2.
Date posted: September 17, 2019.
- Study the photographs labeled K and L which were obtained from the same plant.(Solved)
Study the photographs labeled K and L which were obtained from the same plant.
 i) Identify the sub-division to which they belong.
ii) Give reasons for your answer.
iii) Identify the reproductive structure represented in K and L.
i) Identify the sub-division to which they belong.
ii) Give reasons for your answer.
iii) Identify the reproductive structure represented in K and L.
Date posted: September 17, 2019.
- Two large and one small pump can fill a swimming pool in 4 hours. One large and three small pumps can also fill the...(Solved)
Two large and one small pump can fill a swimming pool in 4 hours. One large and three small pumps can also fill the swimming pool in 4 hours. How many hours will it take 4 large and 4 small pumps.
Date posted: September 17, 2019.
- Which part of the brain regulates body temperatures in mammals?.(Solved)
Which part of the brain regulates body temperatures in mammals?.
Date posted: September 17, 2019.
- Other than cost and ability to conduct, give two other reasons why aluminum is used for making overhead electric cables while magnesium is not.(Solved)
Other than cost and ability to conduct, give two other reasons why aluminum is used for making overhead electric cables while magnesium is not.
Date posted: September 17, 2019.
- The diagram below represents a human arm. Study it and answer the questions that follow.(Solved)
The diagram below represents a human arm. Study it and answer the questions that follow.
 a) Name bone T and R
b) Name muscle C and D
c) i) Identify the fluid found at joint 2.
i) State the function of the fluid in (i) above.
d) i) What is the type of the joint found at part labeled 1.
ii) Differentiate between a tendon and a ligament.
a) Name bone T and R
b) Name muscle C and D
c) i) Identify the fluid found at joint 2.
i) State the function of the fluid in (i) above.
d) i) What is the type of the joint found at part labeled 1.
ii) Differentiate between a tendon and a ligament.
Date posted: September 17, 2019.
- A quantity D is directly proportional to Y and inversely proportional to the square of X. if Y is double and X is increased...(Solved)
A quantity D is directly proportional to Y and inversely proportional to the square of X. if Y is double and X is increased by 20%, find the ratio of the new value of D to the original value of D in the form a:b where a and b are integers.
Date posted: September 17, 2019.
- If 100cm3 of 0.05M hydrogen peroxide were used to produce gas M. calculate the volume of gas M produced if the hydrogen peroxide decomposed completely.(Solved)
If 100cm3 of 0.05M hydrogen peroxide were used to produce gas M. calculate the volume of gas M produced if the hydrogen peroxide decomposed completely.
Date posted: September 17, 2019.
- Determine the equation of the tangent to the curve at the point x = 2. (Solved)
Determine the equation of the tangent to the curve 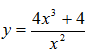 at the point x = 2.
at the point x = 2.
Date posted: September 17, 2019.
- In an experiment, Euglena was put in a petri dish. One side of the petri dish was illuminated and the other kept dark as shown...(Solved)
In an experiment, Euglena was put in a petri dish. One side of the petri dish was illuminated and the other kept dark as shown below.
 a) Name this type of response.
b) State the significance of this type of response in an organism.
c) Other than light, outline other two factors that may cause change of position in Euglena and state the respective type of response.
d) If the above experiment was repeated using a young potted seedling, name the type of response which will be observed.
e) Explain the behavior of the seedling after 3 days.
a) Name this type of response.
b) State the significance of this type of response in an organism.
c) Other than light, outline other two factors that may cause change of position in Euglena and state the respective type of response.
d) If the above experiment was repeated using a young potted seedling, name the type of response which will be observed.
e) Explain the behavior of the seedling after 3 days.
Date posted: September 17, 2019.
- A student set up the apparatus as shown in the diagram below to prepare and collect dry ammonia gas.(Solved)
A student set up the apparatus as shown in the diagram below to prepare and collect dry ammonia gas.
 (i) Identify one mistake in the set-up and give a reason.
(ii) Name a suitable drying agent for ammonia.
(iii) Write an operation for the reaction that occurred when a mixture of ammonium chloride and calcium hydroxide was heated.
(iv) Describe one chemical test for ammonia gas.
(i) Identify one mistake in the set-up and give a reason.
(ii) Name a suitable drying agent for ammonia.
(iii) Write an operation for the reaction that occurred when a mixture of ammonium chloride and calcium hydroxide was heated.
(iv) Describe one chemical test for ammonia gas.
Date posted: September 17, 2019.
- Study the reaction scheme below and answer the questions that follow.(Solved)
Study the reaction scheme below and answer the questions that follow.
 a) Name; Gas M
Precipitate w
b) Write an equation for the formation of ;
The white solid
Colorless solution X
c) What name is given to the process that produce precipitate W
d) State two commercial uses of gas M
a) Name; Gas M
Precipitate w
b) Write an equation for the formation of ;
The white solid
Colorless solution X
c) What name is given to the process that produce precipitate W
d) State two commercial uses of gas M
Date posted: September 17, 2019.
- In the figure above OC is the tangent to the circle. If OE =8cm and OC = 6cm. find EA.(Solved)
In the figure above OC is the tangent to the circle. If OE =8cm and OC = 6cm. find EA.

Date posted: September 17, 2019.
- Below is a simplified diagram of the Downs cell in which sodium metal is manufactured.(Solved)
Below is a simplified diagram of the Downs cell in which sodium metal is manufactured.
 (i) Identify electrolyte X and gas Y.
(ii)Give two properties of sodium that make it possible to collect as shown above.
(iii) Why is graphite preferred to steel as the anode?
(iv) Explain how sodium hydroxide pellets would be obtained from sodium amalgam?
(i) Identify electrolyte X and gas Y.
(ii)Give two properties of sodium that make it possible to collect as shown above.
(iii) Why is graphite preferred to steel as the anode?
(iv) Explain how sodium hydroxide pellets would be obtained from sodium amalgam?
Date posted: September 17, 2019.
- A is matrix and I is the 2 x 2 identity matrix. Determine the values of h and k for which
A-1...(Solved)
A is matrix  and I is the 2 x 2 identity matrix. Determine the values of h and k for which
A-1 = hl + kA.
and I is the 2 x 2 identity matrix. Determine the values of h and k for which
A-1 = hl + kA.
Date posted: September 17, 2019.
- Plants with red flowers were crossed with plants with white flowers. The resultant F1 generation had pink flowers.(Solved)
Plants with red flowers were crossed with plants with white flowers. The resultant F1 generation had pink flowers.Plants with red flowers were crossed with plants with white flowers. The resultant F1 generation had pink flowers.
a) Using R for red flowers and W for white flowers, work out the genotypes of F2 generation.
b) Determine the genotypic ratio of F2 generation.
c) Explain why a cross between red flowered plants and white flowered plants produced pink flowers.
d) If the total number of F2 generation offsprings was 7324. Calculate the number of red flowered plants.
Date posted: September 17, 2019.
- A circle centre is the point C(2,3) passes through a point P(a,b). a point is the mid-point of the line CP.
a)Calculate the coordinates...(Solved)
A circle centre is the point C(2,3) passes through a point P(a,b). a point 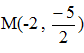 is the mid-point of the line CP.
a)Calculate the coordinates of P.
b)Determine the equation of the circle in the form x2 +y2 +ax + by + c = 0
is the mid-point of the line CP.
a)Calculate the coordinates of P.
b)Determine the equation of the circle in the form x2 +y2 +ax + by + c = 0
Date posted: September 17, 2019.
- The grid below represents part of the periodic table. Study it and answer the questions that follow.
The letters do not represent the actual symbols of...(Solved)
The grid below represents part of the periodic table. Study it and answer the questions that follow.
The letters do not represent the actual symbols of the elements.
 (i) Which element will require the least amount of energy to remove one of its outermost electrons, explain.
(ii) Select the most reactive non-metal
(iii) What name is given to the family of elements to which T belong?
(iv) Between S and R, which element has a smaller atomic radius, explain.
(v) Which of the elements have a tendency of forming covalent bonds? Explain.
(vi) Write the formula of the compound formed when U reacts with T, state the type of body formed.
Formula
Bond
(i) Which element will require the least amount of energy to remove one of its outermost electrons, explain.
(ii) Select the most reactive non-metal
(iii) What name is given to the family of elements to which T belong?
(iv) Between S and R, which element has a smaller atomic radius, explain.
(v) Which of the elements have a tendency of forming covalent bonds? Explain.
(vi) Write the formula of the compound formed when U reacts with T, state the type of body formed.
Formula
Bond
Date posted: September 17, 2019.
- The scheme below shows various reactions and process for some organic compounds.
Study it and answer the questions that follow.(Solved)
The scheme below shows various reactions and process for some organic compounds.
Study it and answer the questions that follow.
 i. Name process I and name any other product formed in the process.
Process
Product
ii) Name the reagent and condition required in process II.
Reagent
Condition
iii) Name the catalyst and reagent required in step IV.
Catalyst
Reagent
iv) Give any one use of the product formed in process (V)
v) Name process (VI)
i. Name process I and name any other product formed in the process.
Process
Product
ii) Name the reagent and condition required in process II.
Reagent
Condition
iii) Name the catalyst and reagent required in step IV.
Catalyst
Reagent
iv) Give any one use of the product formed in process (V)
v) Name process (VI)
Date posted: September 17, 2019.
- a) Expand and simplify (2x + y)5
b) Using the first four terms of the expansion to evaluate 6.025(Solved)
a) Expand and simplify (2x + y)5
b) Using the first four terms of the expansion to evaluate 6.025
Date posted: September 17, 2019.
- a)An organic compound G whose empirical formula is HCO2 has molar mass of 90. It reacts with ethanoic in the presence of a few drops...(Solved)
a)An organic compound G whose empirical formula is HCO2 has molar mass of 90. It reacts with ethanoic in the presence of a few drops of concentrated Sulphuric (VI) acid forming another compound J with a pleasant smell.
(i) Determine the molecular formula of compound G
ii)To which class of homologous series do compound G and J belong?
G
J
b)
i. What is vulcanization of rubber?
i). State two properties of vulcanized rubber
Date posted: September 17, 2019.
- Use the standard electrode potentials given below to answer the questions that follow.(Solved)
Use the standard electrode potentials given below to answer the questions that follow.
 a) Select two half - cells which when combined will give the lowest e.m.f
b) Can a solution of silver nitrate be stored in a container of Zinc?
c) An iron jug was electroplated using Chromium. The chromium electrode and iron jug were thoroughly cleaned and weighed before being dipped into the electrolyte. Why was cleaning necessary?
d) A current of 0.75 Amperes was passed through the solution for one hour and four minutes. The mass of chromium deposited on the jug was 0.52g
(1Faraday = 96500C Cr = 52)
(i) Calculate the quantity of electricity passed.
(ii) How many moles in Chromium were deposited?
(iii) Calculate the quantity of electricity in coulombs required to deposit one mole of Chromium.
(iv) Deduce the charge of the Chromium ion.
a) Select two half - cells which when combined will give the lowest e.m.f
b) Can a solution of silver nitrate be stored in a container of Zinc?
c) An iron jug was electroplated using Chromium. The chromium electrode and iron jug were thoroughly cleaned and weighed before being dipped into the electrolyte. Why was cleaning necessary?
d) A current of 0.75 Amperes was passed through the solution for one hour and four minutes. The mass of chromium deposited on the jug was 0.52g
(1Faraday = 96500C Cr = 52)
(i) Calculate the quantity of electricity passed.
(ii) How many moles in Chromium were deposited?
(iii) Calculate the quantity of electricity in coulombs required to deposit one mole of Chromium.
(iv) Deduce the charge of the Chromium ion.
Date posted: September 17, 2019.
- Evaluate 5 cos2? + 2 = 3sin2? - 2cos ? given that (Solved)
Evaluate 5 cos2θ + 2 = 3sin2θ - 2cos θ given that 
Date posted: September 17, 2019.