 Get premium membership
Get premium membership and access questions with answers, video lessons as well as revision papers.
- 120cm3 of nitrogen gas diffuses through a membrane in forty seconds. How long will it take 240cm3 of carbon (IV) oxide to diffuse through the same membrane (C=12,N=14,O=16)
120cm3 of nitrogen gas diffuses through a membrane in forty seconds. How long will it take 240cm3 of carbon (IV) oxide to diffuse through the same membrane (C=12,N=14,O=16)
Date posted: March 5, 2019
- 100cm³ of 2M nitric acid reacted with 12.5g of a carbonate of metal M. (MCO3) according to the
following equation
100cm³ of 2M nitric acid reacted with 12.5g of a carbonate of metal M. (MCO3) according to the
following equation
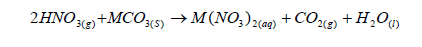
a).Calculate the number of moles of MCO3 that reacted with nitric acid.
b).Calculate the relative molecular mass of MCO3.
Date posted: March 5, 2019
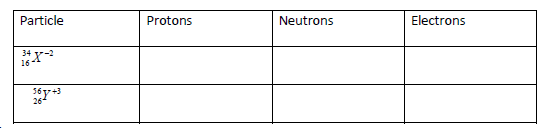
- Complete the table below to show the number of the sub-atomic particles of the given nuclides. X and Y are not actual chemical symbols.
Complete the table below to show the number of the sub-atomic particles of the given nuclides. X and Y are not actual chemical symbols.
Date posted: March 5, 2019
- An organic compound J has the following percentage by mass, carbon, 64.86%, hydrogen, 13.51% and
the rest oxygen. The relative molecular mass of the compound is 74.
An organic compound J has the following percentage by mass, carbon, 64.86%, hydrogen, 13.51% and
the rest oxygen. The relative molecular mass of the compound is 74. Work out the molecular formula of
compound J. [C=12. H=1 O=16]
Date posted: March 5, 2019
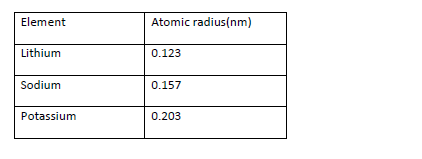
- Use the information in the table about group 1 elements to answer the questions that follow
Use the information in the table about group 1 elements to answer the questions that follow
When the group 1 elements react with water, hydrogen gas is given off. The diagram shows the reaction
of the above three elements with water
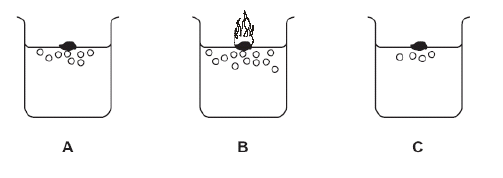
a) What is the general name of the group one elements
b) Which one of these elements A, B or C is Lithium
Date posted: March 5, 2019
- A rectangle whose area is 96m2 is such that its length is 4m longer than its width.
A rectangle whose area is 96m2 is such that its length is 4m longer than its width. Find
a) Its dimensions.
b) Its perimeter.
Date posted: March 5, 2019
- A straight line passes through points A (-2, 6) and B (4, 2).M is the midpoint of line AB. Find the
co-ordinates of M.
A straight line passes through points A (-2, 6) and B (4, 2).M is the midpoint of line AB. Find the
co-ordinates of M.
Date posted: March 5, 2019
- Oketch sells his car to Jane and makes a profit of 17%. Jane sells the same car to Issa at Sh.300,000, making a loss of 10%.Determine the price at which Oketch bought the car.
Oketch sells his car to Jane and makes a profit of 17%. Jane sells the same car to Issa at Sh.300,000, making a loss of 10%.Determine the price at which Oketch bought the car.
Date posted: March 5, 2019
- The 2nd, 4th and 7th terms of an AP are the first 3 consecutive terms of a GP. If the common difference of the AP is 2 , find the common ratio of the G.p
The 2nd, 4th and 7th terms of an AP are the first 3 consecutive terms of a GP. If the common difference of the AP is 2 , find the common ratio of the G.p
Date posted: March 5, 2019
- In a book store, books packed in cartons are arranged in rows such that there are 50 cartons in the first row, 48 cartons in the next row, 46 in the next and so on.
In a book store, books packed in cartons are arranged in rows such that there are 50 cartons in the first row, 48 cartons in the next row, 46 in the next and so on.
a) How many cartons will be there in the 8th row?
b) If there are 20 rows in total, find total number of cartons in the book store.
Date posted: March 5, 2019
- Find the values of x in the question below
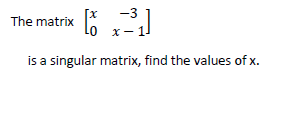
Date posted: March 5, 2019
- Find the gradient of y=x2 -3x at x=1
Find the gradient of y=x2 -3x at x=1
Date posted: March 5, 2019
- A bus and a car leave Nairobi at 7.30am and 9.30am respectively. If their speeds are 60km/h and 100km/h respectively, find the time when the car catches up with the bus.
A bus and a car leave Nairobi at 7.30am and 9.30am respectively. If their speeds are 60km/h and 100km/h respectively, find the time when the car catches up with the bus.
Date posted: March 5, 2019
- Solve the inequality below and state the integral values of your solution.
Solve the inequality below and state the integral values of your solution.
2(3x-4)
Date posted: March 5, 2019
- In 100m race there are three main competitors namely Simiyu, Ondiek and Kamau. Simiyu is three times likely to win as Ondiek, while Ondiek is twice as likely to win as Kamau.
In 100m race there are three main competitors namely Simiyu, Ondiek and Kamau. Simiyu is three times likely to win as Ondiek, while Ondiek is twice as likely to win as Kamau.
Find the probability that
a) Ondiek wins the race
b) Either Simiyu or Kamau
Date posted: March 5, 2019
- An electric crane lifts a load of 2000kg through a vertical distance of 3.0m in 6 seconds
Determine:
An electric crane lifts a load of 2000kg through a vertical distance of 3.0m in 6 seconds
Determine:
i)The work done
ii) The power developed by the crane.
iii).The efficiency of the crane if it is operated by an electric motor rated 12.5kw.
Date posted: March 5, 2019
- In an experiment to determine the density of solid S which is not soluble in water. A student obtained the following;
In an experiment to determine the density of solid S which is not soluble in water. A student obtained the following;
- Mass of empty density bottle= 20g
- Mass of density bottle when full of water =45g
- Mass of density bottle with small quantity of solid S =152g
- Mass of density bottle with small amount of solids S and topped up with water =167g
Given that the density of water is 1g/cm3,
Find;
i.The volume of the density bottle
ii. The mass of the solid S
iii. The volume of the solid S
Date posted: March 5, 2019
- The level of water in a measuring cylinder rises from the 50cm3 mark to 55.7cm3 marks when a metal block weighing 45g is submerged in the water in the cylinder.Calculate the density of the metal block.
The level of water in a measuring cylinder rises from the 50cm3 mark to 55.7cm3 marks when a metal block weighing 45g is submerged in the water in the cylinder.Calculate the density of the metal block.
Date posted: March 5, 2019
- A girl ties a stone of mass 250g with a piece of thread. Holding the other end of the thread at 45cm in length, she makes the stone revolve in a circle in a vertical plane at a constant speed.
A girl ties a stone of mass 250g with a piece of thread. Holding the other end of the thread at 45cm in length, she makes the stone revolve in a circle in a vertical plane at a constant speed. If the stone is making 3 revolutions, per second calculate;
a) The angular velocity.
b) Tension in the string when the stone is vertically above the girl’s hand.
Date posted: March 5, 2019
- An oil drop forms a circular patch of an area 5.0 x 10-13cm2. If the drop has a volume of 9.0 x 10-14 cm3, estimate the diameter of the oil molecule.
An oil drop forms a circular patch of an area 5.0 x 10-13cm2. If the drop has a volume of 9.0 x 10-14 cm3, estimate the diameter of the oil molecule.
Date posted: March 5, 2019