 Get premium membership
Get premium membership and access questions with answers, video lessons as well as revision papers.
- Factorize completely; a2-15ab +36b2
Factorize completely; a2-15ab +36b2
Date posted: February 23, 2019
- 1.60g of an oxide of Magnesium contain 0.84g by mass of Magnesium. Determine its empirical formula(Mg = 24.0, 16.0)
1.60g of an oxide of Magnesium contain 0.84g by mass of Magnesium. Determine its empirical formula(Mg = 24.0, 16.0)
Date posted: February 25, 2019
- Solve for the following
27x +33x+1 =108
Solve for the following
27x +33x+1 =108
Date posted: February 25, 2019
- Solve the simultaneous equation
3x – 4y =10
X + 3y = -1
Solve the simultaneous equation
3x – 4y =10
X + 3y = -1
Date posted: February 26, 2019
- Find the value of x in
(logx)2 -15(logx) + 56 = 0
Find the value of x in
(logx)2 -15(logx) + 56 = 0
Date posted: February 26, 2019
- Find the equation of a line passing through (4, -3) and parallel to the line whose equation is
4y – 3x = 8.
Find the equation of a line passing through (4, -3) and parallel to the line whose equation is
4y – 3x = 8.
Date posted: February 26, 2019
- Simplify the equation below
Simplify the equation below
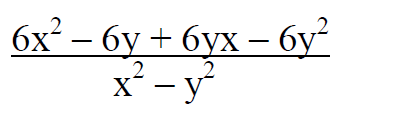
Date posted: February 26, 2019
- Solve for X in the equation below 32(x - 3) ÷ 8(x - 4) = 64 ÷ 2x
Solve for X in the equation below
32(x - 3) ÷ 8(x - 4) = 64 ÷ 2x
Date posted: February 26, 2019
- Given that a=2, b=1, and c=3, find the value of
Given that a=2, b=1, and c=3, find the value of
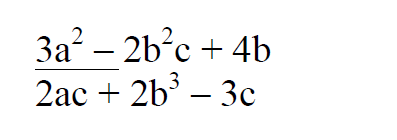
Date posted: February 26, 2019
- Arrange the following fractions in descending order.
Arrange the following fractions in descending order.
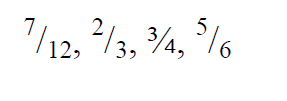
Date posted: February 26, 2019
- The cost of a pen is sh.X and that of a pencil is sh. 4 less than that of a pen. Find the combined cost of 4 pens and 5 pencils.
The cost of a pen is sh.X and that of a pencil is sh. 4 less than that of a pen. Find the combined cost of 4 pens and 5 pencils.
Date posted: February 26, 2019
- Factorize: 6x2 -13x + 6
Factorize: 6x2 -13x + 6
Date posted: February 26, 2019
- During heating of a hydrated copper (II)sulphate crystals, the following readings were obtained:
Mass of evaporating dish =300.0g, Mass of evaporating dish + hydrated salt = 305.0g, Mass of evaporating dish + anhydrous salt = 303.2g
During heating of a hydrated copper (II)sulphate crystals, the following readings were obtained:
Mass of evaporating dish =300.0g, Mass of evaporating dish + hydrated salt = 305.0g, Mass of evaporating dish + anhydrous salt = 303.2g
Calculate the number of water of crystallization molecules in hydrated copper (II)sulphate.(Cu =64.5, S = 32.0,O=16.0, H = 1.0)
Date posted: February 26, 2019
- An element Q consists of 3 isotopes of mass 28, 29, 30 and percentage abundance of 92.2%, 4.7%, 3.1% respectively. Determine the relative atomic mass of the element?
An element Q consists of 3 isotopes of mass 28, 29, 30 and percentage abundance of 92.2%, 4.7%, 3.1% respectively. Determine the relative atomic mass of the element?
Date posted: February 26, 2019
- An element N forms an ion N2. This ion contains 18 electrons:
a) State the period and the group of the periodic table that element N belongs.
An element N forms an ion N2. This ion contains 18 electrons:
a) State the period and the group of the periodic table that element N belongs.
b) Write the formula of the compound which would be formed if N reacts with bromine.
Date posted: February 26, 2019
- When 27.8g of hydrated aluminium oxide, Al2O3.xH2O, was heated to a constant mass, 20.6g of aluminium oxide was obtained. Determine the value of x (H=1, O=16, A=27)
When 27.8g of hydrated aluminium oxide, Al2O3.xH2O, was heated to a constant mass, 20.6g of aluminium oxide was obtained. Determine the value of x (H=1, O=16, A=27)
Date posted: February 26, 2019
- 6g of iron reacted with hydrogen chloride gas to form iron (II) chloride at S.T.P. Calculate the volume of the hydrogen chloride
gas used. (Fe=56, molar gas volume at S.T.P=22.4dm3 )
6g of iron reacted with hydrogen chloride gas to form iron (II) chloride at S.T.P. Calculate the volume of the hydrogen chloride
gas used. (Fe=56, molar gas volume at S.T.P=22.4dm3 )
Date posted: February 26, 2019
- Study the information in the table below and answer the questions that follow.
Study the information in the table below and answer the questions that follow.
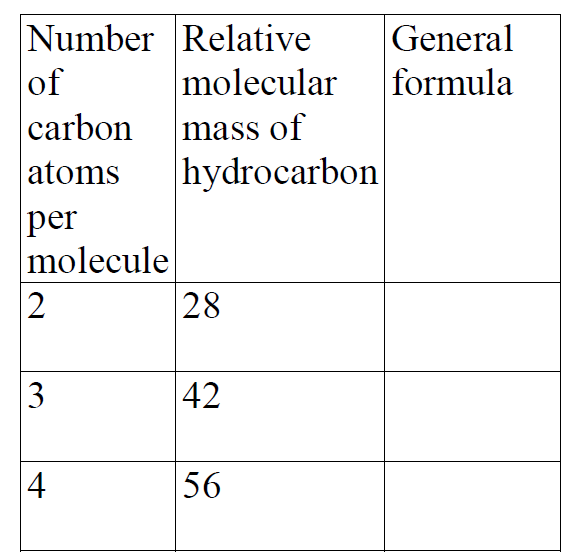
i) Write the general formula of the hydrocarbons in the table.
ii) Predict the relative molecular mass of the hydrocarbon with 5 carbon atoms.
iii) Determine the molecular formula of the hydrocarbon in (II) above and draw its structural formula (H=1.0, C = 12.0)
Date posted: February 26, 2019
- An organic compound contains 24.24% carbon 4.04% hydrogen and the rest is chlorine
a) Find its empirical formula. (C=12, H=1, Cl=35.5)
b) Find its molecular formula if its R.M.M is 99.
An organic compound contains 24.24% carbon 4.04% hydrogen and the rest is chlorine
a) Find its empirical formula. (C=12, H=1, Cl=35.5)
b) Find its molecular formula if its R.M.M is 99.
Date posted: February 26, 2019
- Using dots (.) and crosses (x) to represent outermost electrons, draw diagrams to show bonding in.
a)Carbon (IV) oxide
b) NH4+
Using dots (.) and crosses (x) to represent outermost electrons, draw diagrams to show bonding in.
a)Carbon (IV) oxide
b) NH4+
Date posted: February 26, 2019