 Get premium membership
Get premium membership and access questions with answers, video lessons as well as revision papers.
- Write an equation to show the effect of heat on:
i) Potassium nitrate, KNO3
ii) silver nitrate, AgNO3
Write an equation to show the effect of heat on:
i) Potassium nitrate, KNO3
ii) silver nitrate, AgNO3
Date posted: February 26, 2019
- Calculate the percentage nitrogen content in the following ammonium salts.
i) Ammonium sulphate (NH4)2SO4
ii) Urea, (NH2)2CO
N= 14, H=1, S=32, O=16, C=12
Calculate the percentage nitrogen content in the following ammonium salts.
i) Ammonium sulphate (NH4)2SO4
ii) Urea, (NH2)2CO
N= 14, H=1, S=32, O=16, C=12
Date posted: February 26, 2019
- 4.0g of magnesium carbonate have some impurity of magnesium sulphate were dissolved in 250cm3 of 1.0m HCL. 25cm3 of the resulting solution was found to require 28.8cm3 of 0.6M sodium hydroxide for complete neutralization.Calculate the number of moles of sodium hydroxide in 28.8cm3.
4.0g of magnesium carbonate have some impurity of magnesium sulphate were dissolved in 250cm3 of 1.0m HCL. 25cm3 of the resulting solution was found to require 28.8cm3 of 0.6M sodium hydroxide for complete neutralization.Calculate the number of moles of sodium hydroxide in 28.8cm3.
Date posted: February 26, 2019
- Draw the structural formulae of the following compounds:
a) 2, 3 – dibromobutane
b) 2, 3, 4 – trimethyl pentene
c) 4 – chloro – 5 – methylhex – 2 –yne
Draw the structural formulae of the following compounds:
a) 2, 3 – dibromobutane
b) 2, 3, 4 – trimethyl pentene
c) 4 – chloro – 5 – methylhex – 2 –yne
Date posted: February 26, 2019
- Balance the chemical equation given below
C3H8 + O2 ?H2O + CO2
Balance the chemical equation given below
C3H8 + O2 ?H2O + CO2
Date posted: February 26, 2019
- 30cm3 of hydrogen chloride gas diffuses through a porous pot in 20 seconds. How long would it take 42cm3 of sulphur(IV) oxide gas to diffuse through the same pot under the same conditions (H =1 Cl = 35.5 S = 32 O =16)
30cm3 of hydrogen chloride gas diffuses through a porous pot in 20 seconds. How long would it take 42cm3 of sulphur(IV) oxide gas to diffuse through the same pot under the same conditions (H =1 Cl = 35.5 S = 32 O =16)
Date posted: February 26, 2019
- Express 10,500 in terms of its prime factors and determine the smallest value of p such that 10,500p is a
perfect cube
Express 10,500 in terms of its prime factors and determine the smallest value of p such that 10,500p is a
perfect cube
Date posted: February 26, 2019
- Find the size of land each child got in the question below

Date posted: February 26, 2019
- Without using tables evaluate
Without using tables evaluate
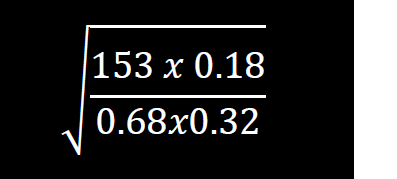
Date posted: February 26, 2019
- The cost of 5 skirts and 3 blouses is sh 1,750. Mueni bought three of the skirts and one of the blouses for sh 850.find the cost of each item.
The cost of 5 skirts and 3 blouses is sh 1,750. Mueni bought three of the skirts and one of the blouses for sh 850.find the cost of each item.
Date posted: February 26, 2019
- Abdi sold a radio costing ksh 3,800 at a profit of 20%.He earned a commission of 22.5% on the profit.Find the amount he earned.
Abdi sold a radio costing ksh 3,800 at a profit of 20%.He earned a commission of 22.5% on the profit.Find the amount he earned.
Date posted: February 26, 2019
- Musa paid sh 180 for a shirt after getting a discount of 10%.The shopkeeper made a profit of 20% on the sale of this shirt.What percentage profit would the shopkeeper have made if no discount was allowed?.
Musa paid sh 180 for a shirt after getting a discount of 10%.The shopkeeper made a profit of 20% on the sale of this shirt.What percentage profit would the shopkeeper have made if no discount was allowed?.
Date posted: February 26, 2019
- The size of each interior angle of a regular polygon is five times the size of its exterior angle.Find the
number of sides of the polygon.
The size of each interior angle of a regular polygon is five times the size of its exterior angle.Find the
number of sides of the polygon.
Date posted: February 26, 2019
- Find the value of x given that: 49x+1 + 7 =350
Find the value of x given that: 49x+1 + 7 =350
Date posted: February 26, 2019
- A perpendicular line is drawn from a point (3,5) to the line 2y+x=3.Find the equation of the perpendicular line.
A perpendicular line is drawn from a point (3,5) to the line 2y+x=3.Find the equation of the perpendicular line.
Date posted: February 26, 2019
- The ratio of the lengths of corresponding sides of two similar rectangular water tanks is 3:5.The volume of the smaller tank is 8.1m3. Calculate the volume of the larger tank.
The ratio of the lengths of corresponding sides of two similar rectangular water tanks is 3:5.The volume of the smaller tank is 8.1m3. Calculate the volume of the larger tank.
Date posted: February 26, 2019
- Solve the question below
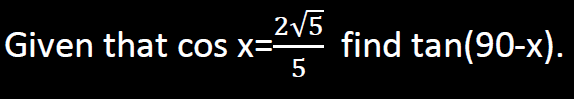
Date posted: February 26, 2019
- Two sides of a triangle are 5 cm each and the angle between them is 1200.Calculate the area of the triangle.
Two sides of a triangle are 5 cm each and the angle between them is 1200.Calculate the area of the triangle.
Date posted: February 26, 2019
- Factorize the question below
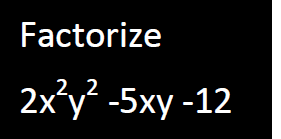
Date posted: February 26, 2019
- Find the range of x if 2= 3-x<5
Find the range of x if 2= 3-x<5
Date posted: February 27, 2019