 Get premium membership
Get premium membership and access questions with answers, video lessons as well as revision papers.
- Express 10,500 in terms of its prime factors and determine the smallest value of p such that 10,500p is a
perfect cube
Express 10,500 in terms of its prime factors and determine the smallest value of p such that 10,500p is a
perfect cube
Date posted: February 26, 2019
- Find the size of land each child got in the question below

Date posted: February 26, 2019
- Without using tables evaluate
Without using tables evaluate
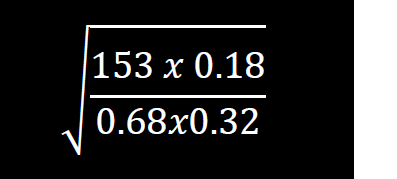
Date posted: February 26, 2019
- The cost of 5 skirts and 3 blouses is sh 1,750. Mueni bought three of the skirts and one of the blouses for sh 850.find the cost of each item.
The cost of 5 skirts and 3 blouses is sh 1,750. Mueni bought three of the skirts and one of the blouses for sh 850.find the cost of each item.
Date posted: February 26, 2019
- Abdi sold a radio costing ksh 3,800 at a profit of 20%.He earned a commission of 22.5% on the profit.Find the amount he earned.
Abdi sold a radio costing ksh 3,800 at a profit of 20%.He earned a commission of 22.5% on the profit.Find the amount he earned.
Date posted: February 26, 2019
- Musa paid sh 180 for a shirt after getting a discount of 10%.The shopkeeper made a profit of 20% on the sale of this shirt.What percentage profit would the shopkeeper have made if no discount was allowed?.
Musa paid sh 180 for a shirt after getting a discount of 10%.The shopkeeper made a profit of 20% on the sale of this shirt.What percentage profit would the shopkeeper have made if no discount was allowed?.
Date posted: February 26, 2019
- The size of each interior angle of a regular polygon is five times the size of its exterior angle.Find the
number of sides of the polygon.
The size of each interior angle of a regular polygon is five times the size of its exterior angle.Find the
number of sides of the polygon.
Date posted: February 26, 2019
- Find the value of x given that: 49x+1 + 7 =350
Find the value of x given that: 49x+1 + 7 =350
Date posted: February 26, 2019
- A perpendicular line is drawn from a point (3,5) to the line 2y+x=3.Find the equation of the perpendicular line.
A perpendicular line is drawn from a point (3,5) to the line 2y+x=3.Find the equation of the perpendicular line.
Date posted: February 26, 2019
- The ratio of the lengths of corresponding sides of two similar rectangular water tanks is 3:5.The volume of the smaller tank is 8.1m3. Calculate the volume of the larger tank.
The ratio of the lengths of corresponding sides of two similar rectangular water tanks is 3:5.The volume of the smaller tank is 8.1m3. Calculate the volume of the larger tank.
Date posted: February 26, 2019
- Solve the question below
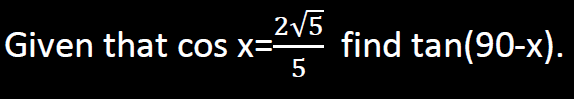
Date posted: February 26, 2019
- Two sides of a triangle are 5 cm each and the angle between them is 1200.Calculate the area of the triangle.
Two sides of a triangle are 5 cm each and the angle between them is 1200.Calculate the area of the triangle.
Date posted: February 26, 2019
- Factorize the question below
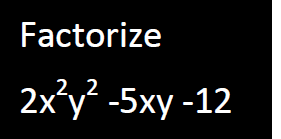
Date posted: February 26, 2019
- Find the range of x if 2= 3-x<5
Find the range of x if 2= 3-x<5
Date posted: February 27, 2019
- Solve the equations below:
X2 + y2 = 26
x-y =-4
Solve the equations below:
X2 + y2 = 26
x-y =-4
Date posted: February 27, 2019
- Find the value of x in;
log(x-1) +2=log (3x+2)+log 25
Find the value of x in;
log(x-1) +2=log (3x+2)+log 25
Date posted: February 27, 2019
- Omondi deposited sh 4500 in a bank which paid compound interest at 12% p.a.Calculate the amount after 2 years.
Omondi deposited sh 4500 in a bank which paid compound interest at 12% p.a.Calculate the amount after 2 years.
Date posted: February 27, 2019
- Make y the subject of the formula
Make y the subject of the formula
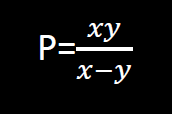
Date posted: February 27, 2019
- The 5th and 10th terms of an A.P are 18 and -2 respectively.Find the first term and the common difference of the A.p.
The 5th and 10th terms of an A.P are 18 and -2 respectively.Find the first term and the common difference of the A.p.
Date posted: February 27, 2019
- Determine the probability in the question below

Date posted: February 27, 2019